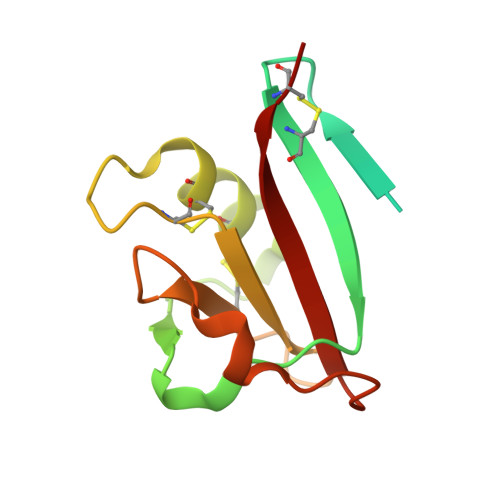
The structure of leech anti-platelet protein, an inhibitor of haemostasis.
Huizinga, E.G., Schouten, A., Connolly, T.M., Kroon, J., Sixma, J.J., Gros, P.(2001) Acta Crystallogr D Biol Crystallogr 57: 1071-1078
- PubMed: 11468390 Search on PubMed
- DOI: https://doi.org/10.1107/s0907444901007405
- Primary Citation Related Structures:
1I8N - PubMed Abstract:
Leech anti-platelet protein (LAPP) from the leech Haementeria officinalis is a collagen-binding protein that inhibits the collagen-mediated adhesion of blood platelets. The crystal structure of recombinant LAPP has been determined using single isomorphous replacement with anomalous scattering combined with solvent flattening and threefold molecular averaging. The model of LAPP has been refined to 2.2 A resolution (R factor 21.5%; free R factor 24.0%). LAPP contains an 89-residue C-terminal domain consisting of a central six-stranded antiparallel beta-sheet flanked on one side by an alpha-helix and on the other side by two extended loops with little secondary structure. A 36-residue N-terminal region is not visible in the electron-density map. This region is rich in glycine and lacks hydrophobic residues. It probably does not have a compact globular fold, but instead has an extended conformation and is flexible. The crystal packing suggests that LAPP may form tightly interacting dimers. The fold of the C-terminal domain of LAPP closely resembles that of the N-domain of hepatocyte growth factor (HGF), which classifies LAPP as a PAN domain. However, no significant sequence homology exists between LAPP and other PAN domains. Common structural features between LAPP and the HGF N-domain include two disulfide bonds that link the alpha-helix to the central region of the protein and five residues with a conserved hydrophobic nature that are located in the core of the domain. These conserved structural features may be an important determinant of the PAN-domain type of fold.
- Thrombosis and Haemostasis Laboratory, Department of Haematology, Institute of Biomembranes, University Medical Center Utrecht, 3508 CH Utrecht, The Netherlands.
Organizational Affiliation: