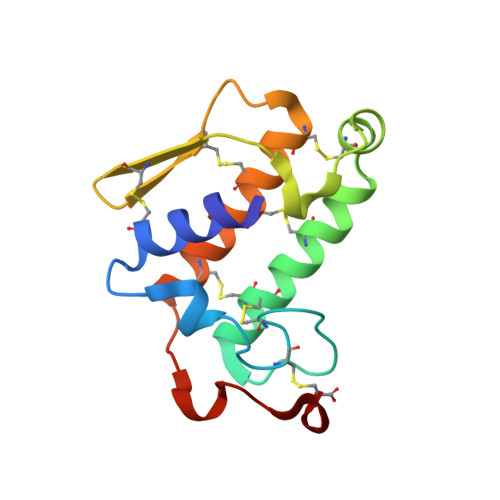
X-ray structure of bovine pancreatic phospholipase A2 at atomic resolution.
Steiner, R.A., Rozeboom, H.J., de Vries, A., Kalk, K.H., Murshudov, G.N., Wilson, K.S., Dijkstra, B.W.(2001) Acta Crystallogr D Biol Crystallogr 57: 516-526
- PubMed: 11264580 Search on PubMed
- DOI: https://doi.org/10.1107/s0907444901002530
- Primary Citation Related Structures:
1G4I - PubMed Abstract:
Using synchrotron radiation and a CCD camera, X-ray data have been collected from wild-type bovine pancreatic phospholipase A(2) at 100 K to 0.97 A resolution allowing full anisotropic refinement. The final model has a conventional R factor of 9.44% for all reflections, with a mean standard uncertainty for the positional parameters of 0.031 A as calculated from inversion of the full positional least-squares matrix. At 0.97 A resolution, bovine pancreatic phospholipase A(2) reveals for the first time that its rigid scaffolding does not preclude flexibility, which probably plays an important role in the catalytic process. Functionally important regions (the interfacial binding site and calcium-binding loop) are located at the molecular surface, where conformational variability is more pronounced. A cluster of 2-methyl-2,4-pentanediol molecules is present at the entrance of the hydrophobic channel that leads to the catalytic site and mimics the fatty-acid chains of a substrate analogue. Bovine pancreatic phospholipase A(2) at atomic resolution is compared with previous crystallographic structures and with models derived from nuclear magnetic resonance studies. Given the high structural similarity among extracellular phospholipases A(2) observed so far at lower resolution, the results arising from this structural analysis are expected to be of general validity for this class of enzymes.
- Laboratory of Biophysical Chemistry, Department of Chemistry, University of Groningen, Nijenborgh 4, 9747 AG Groningen, The Netherlands.
Organizational Affiliation: