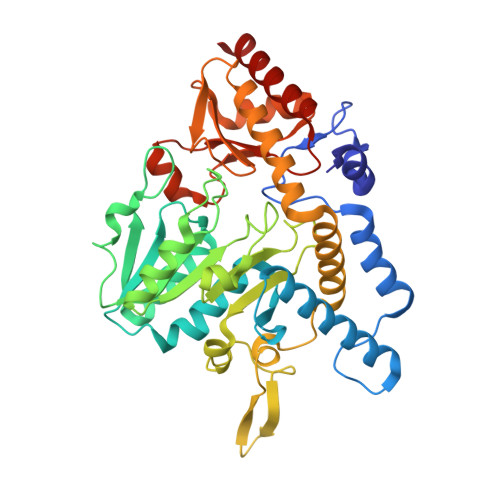
Structure of a NifS homologue: X-ray structure analysis of CsdB, an Escherichia coli counterpart of mammalian selenocysteine lyase
Fujii, T., Maeda, M., Mihara, H., Kurihara, T., Esaki, N., Hata, Y.(2000) Biochemistry 39: 1263-1273
- PubMed: 10684605
- DOI: https://doi.org/10.1021/bi991732a
- Primary Citation of Related Structures:
1C0N - PubMed Abstract:
Escherichia coli CsdB, a NifS homologue with a high specificity for L-selenocysteine, is a pyridoxal 5'-phosphate (PLP)-dependent dimeric enzyme that belongs to aminotransferases class V in fold-type I of PLP enzymes and catalyzes the decomposition of L-selenocysteine into selenium and L-alanine. The crystal structure of the enzyme has been determined by the X-ray crystallographic method of multiple isomorphous replacement and refined to an R-factor of 18.7% at 2.8 A resolution. The subunit structure consists of three parts: a large domain of an alpha/beta-fold containing a seven-stranded beta-sheet flanked by seven helices, a small domain containing a four-stranded antiparallel beta-sheet flanked by three alpha-helices, and an N-terminal segment containing two alpha-helices. The overall fold of the subunit is similar to those of the enzymes belonging to the fold-type I family represented by aspartate aminotransferase. However, CsdB has several structural features that are not observed in other families of the enzymes. A remarkable feature is that an alpha-helix in the lobe extending from the small domain to the large domain in one subunit of the dimer interacts with a beta-hairpin loop protruding from the large domain of the other subunit. The extended lobe and the protruded beta-hairpin loop form one side of a limb of each active site in the enzyme. The most striking structural feature of CsdB lies in the location of a putative catalytic residue; the side chain of Cys364 on the extended lobe of one subunit is close enough to interact with the gamma-atom of a modeled substrate in the active site of the subunit. Moreover, His55 from the other subunit is positioned so that it interacts with the gamma- or beta-atom of the substrate and may be involved in the catalytic reaction. This is the first report on three-dimensional structures of NifS homologues.
- Institute for Chemical Research, Kyoto University, Uji, Kyoto 611-0011, Japan.
Organizational Affiliation: