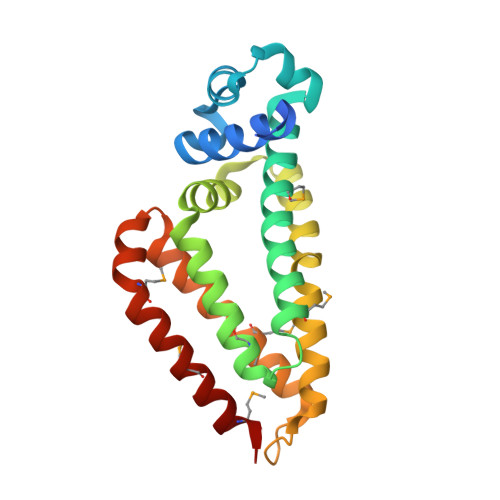
Crystal structure of TM1030 from Thermotoga maritima at 2.3 A resolution reveals molecular details of its transcription repressor function.
Premkumar, L., Rife, C.L., Sri Krishna, S., McMullan, D., Miller, M.D., Abdubek, P., Ambing, E., Astakhova, T., Axelrod, H.L., Canaves, J.M., Carlton, D., Chiu, H.J., Clayton, T., DiDonato, M., Duan, L., Elsliger, M.A., Feuerhelm, J., Floyd, R., Grzechnik, S.K., Hale, J., Hampton, E., Han, G.W., Haugen, J., Jaroszewski, L., Jin, K.K., Klock, H.E., Knuth, M.W., Koesema, E., Kovarik, J.S., Kreusch, A., Levin, I., McPhillips, T.M., Morse, A.T., Nigoghossian, E., Okach, L., Oommachen, S., Paulsen, J., Quijano, K., Reyes, R., Rezezadeh, F., Rodionov, D., Schwarzenbacher, R., Spraggon, G., van den Bedem, H., White, A., Wolf, G., Xu, Q., Hodgson, K.O., Wooley, J., Deacon, A.M., Godzik, A., Lesley, S.A., Wilson, I.A.(2007) Proteins 68: 418-424
- PubMed: 17444523 Search on PubMed
- DOI: https://doi.org/10.1002/prot.21436
- Primary Citation Related Structures:
1ZKG - Burnham Institute for Medical Research, La Jolla, California, USA.
Organizational Affiliation: