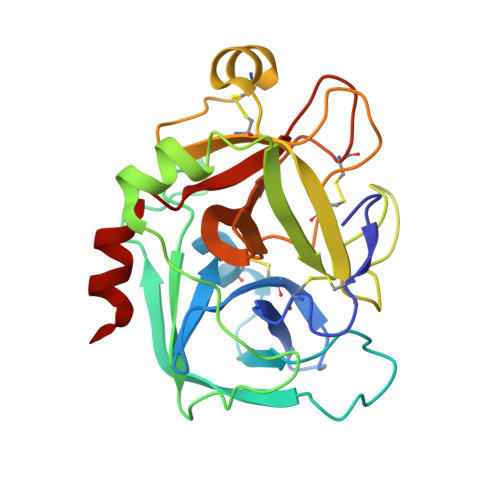
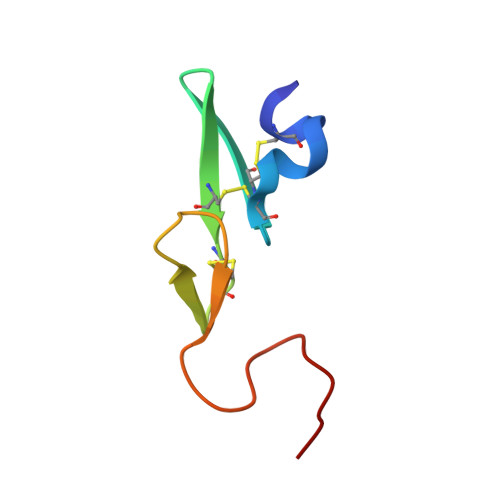
Discovery of 1-(3'-aminobenzisoxazol-5'-yl)-3-trifluoromethyl-N-[2-fluoro-4-[(2'-dimethylaminomethyl)imidazol-1-yl]phenyl]-1H-pyrazole-5-carboxyamide hydrochloride (razaxaban), a highly potent, selective, and orally bioavailable factor Xa inhibitor.
Quan, M.L., Lam, P.Y.S., Han, Q., Pinto, D.J.P., He, M.Y., Li, R., Ellis, C.D., Clark, C.G., Teleha, C.A., Sun, J.-H., Alexander, R.S., Bai, S., Luettgen, J.M., Knabb, R.M., Wong, P.C., Wexler, R.R.(2005) J Med Chem 48: 1729-1744
- PubMed: 15771420
- DOI: https://doi.org/10.1021/jm0497949
- Primary Citation Related Structures:
1Z6E - PubMed Abstract:
Modification of a series of pyrazole factor Xa inhibitors to incorporate an aminobenzisoxazole as the P(1) ligand resulted in compounds with improved selectivity for factor Xa relative to trypsin and plasma kallikrein. Further optimization of the P(4) moiety led to compounds with enhanced permeability and reduced protein binding. The SAR and pharmacokinetic profile of this series of compounds is described herein. These efforts culminated in 1-(3'-aminobenzisoxazol-5'-yl)-3-trifluoromethyl-N-[2-fluoro-4-[(2'-dimethylaminomethyl)imidazol-1-yl]phenyl]-1H-pyrazole-5-carboxyamide (11d), a potent, selective, and orally bioavailable inhibitor of factor Xa. On the basis of its excellent in vitro potency and selectivity profile, high free fraction in human plasma, good oral bioavailability, and in vivo efficacy in antithrombotic models, the HCl salt of this compound was selected for clinical development as razaxaban (DPC 906, BMS-561389).
- Discovery Chemistry, Pharmaceutical Research Institute, Bristol-Myers Squibb Co., P.O. Box 5400, Princeton, New Jersey 08543-5400, USA. mimi.quan@bms.com
Organizational Affiliation: