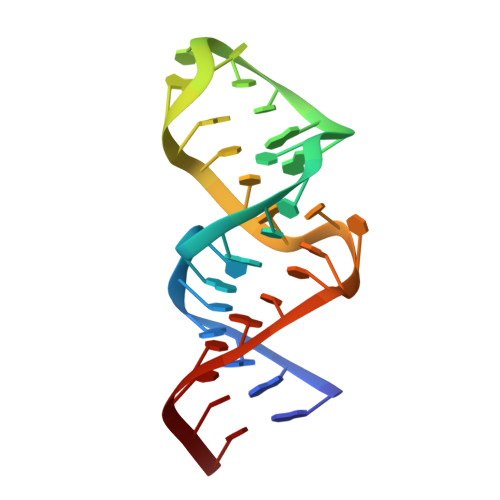
The structure of an enzyme-activating fragment of human telomerase RNA.
Leeper, T.C., Varani, G.(2005) RNA 11: 394-403
- PubMed: 15703438 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1261/rna.7222505
- Primary Citation Related Structures:
1Z31 - PubMed Abstract:
The ribonucleoprotein enzyme telomerase ensures the stability and fidelity of linear chromosome ends by elongating the telomeric DNA that is lost during each round of DNA replication. All telomerases contain a catalytic protein component homologous to viral reverse transcriptases (TERT) and an RNA (TR) that provides the template sequence, acts as the scaffold for ribonucleoprotein assembly, and activates the enzyme for catalysis. Vertebrate telomerase RNAs contain three highly conserved structural and functional domains: the template domain, the "CR4-CR5" or "activation" domain essential for activation of the enzymatic activity, and a 3'-terminal "box H/ACA"-homology domain responsible for ribonucleprotein assembly and maturation. Here we report the NMR structure of a functionally essential RNA structural element derived from the human telomerase RNA CR4-CR5 domain. This RNA, referred to as hTR J6, forms a stable hairpin interrupted by a single nucleotide bulge and an asymmetric internal loop. Previous work on telomerase has shown that deletion of the hTR J6 asymmetric internal loop results in an RNA incapable of binding the enzymatic protein component of the RNP and therefore an inactive RNP without telomerase activity. We demonstrate here that the J6 internal loop introduces a twist in the RNA structure that may position the entire domain into the catalytic site of the enzyme.
- Department of Biochemistry and Department of Chemistry, University of Washington, Seattle, WA 98195-1700, USA.
Organizational Affiliation: