ERbeta ligands. 3. Exploiting two binding orientations of the 2-phenylnaphthalene scaffold to achieve ERbeta selectivity
Mewshaw, R.E., Edsall Jr., R.J., Yang, C., Manas, E.S., Xu, Z.B., Henderson, R.A., Keith Jr., J.C., Harris, H.A.(2005) J Med Chem 48: 3953-3979
- PubMed: 15943471 Search on PubMed
- DOI: https://doi.org/10.1021/jm058173s
- Primary Citation Related Structures:
1YY4, 1YYE - PubMed Abstract:
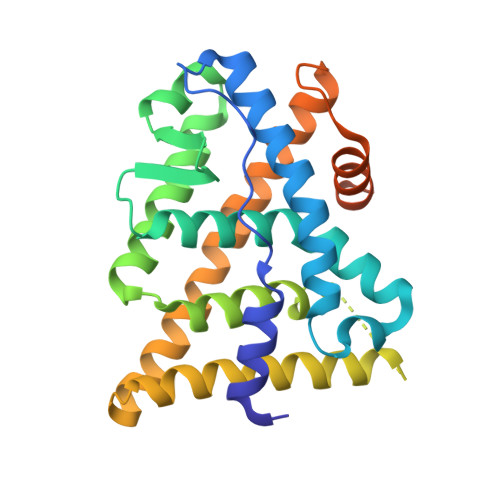
The 2-phenylnaphthalene scaffold was explored as a simplified version of genistein in order to identify ER selective ligands. With the aid of docking studies, positions 1, 4, and 8 of the 2-phenylnaphthalene template were predicted to be the most potentially influential positions to enhance ER selectivity using two different binding orientations. Both orientations have the phenol moiety mimicking the A-ring of genistein. Several compounds predicted to adopt orientations similar to that of genistein when bound to ERbeta were observed to have slightly higher ER affinity and selectivity than genistein. The second orientation we exploited, which was different from that of genistein when bound to ERbeta, resulted in the discovery of several compounds that had superior ER selectivity and affinity versus genistein. X-ray structures of two ER selective compounds (i.e., 15 and 47) confirmed the alternate binding mode and suggested that substituents at positions 1 and 8 were responsible for inducing selectivity. One compound (i.e., 47, WAY-202196) was further examined and found to be effective in two models of inflammation, suggesting that targeting ER may be therapeutically useful in treating certain chronic inflammatory diseases.
- Chemical and Screening Sciences, Wyeth Research, Collegeville, Pennsylvania 19426, USA. mewshar@wyeth.com
Organizational Affiliation: