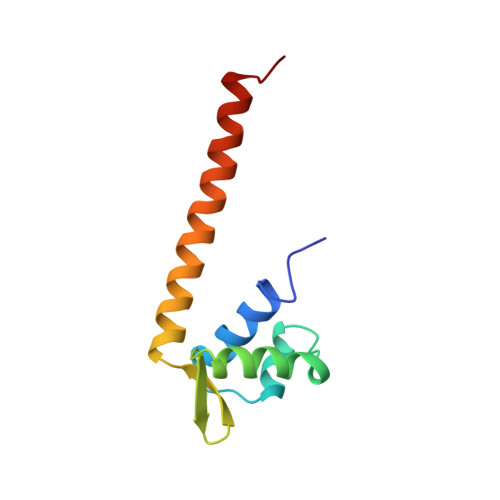
Solution Structure, Stability, and Flexibility of Sso10a: A Hyperthermophile Coiled-Coil DNA-Binding Protein(,).
Kahsai, M.A., Vogler, B., Clark, A.T., Edmondson, S.P., Shriver, J.W.(2005) Biochemistry 44: 2822-2832
- PubMed: 15723526
- DOI: https://doi.org/10.1021/bi047669t
- Primary Citation of Related Structures:
1XSX - PubMed Abstract:
Sso10a is one of a number of DNA-binding proteins from the hyperthermophile Sulfolobus solfataricus that has been associated with DNA packaging and chromatin regulation. Sequence analysis indicates that it is a member of a conserved group of archaeal transcription regulators (COG3432). We have determined the solution structure of Sso10a and show that it is a homodimer of winged-helix DNA-binding domains. The dimer interface consists of an extended antiparallel coiled coil, with the globular DNA-binding domains positioned at opposite ends of a solvent-exposed coiled-coil rod. NMR structure refinement of the elongated structure benefited not only from the inclusion of residual dipolar couplings from partially aligned samples but also the influence of anisotropic rotational diffusion on heteronuclear relaxation. An analysis of backbone mobility using (15)N relaxation rates indicated that the overall tertiary and quaternary structure is largely inflexible on the nanosecond to picosecond time scale. Amide hydrogen exchange data demonstrated that the most stable region of the protein extends from the core of the winged helices into the coiled coil. The positions of the globular heads relative to the coiled coil in solution deviate only slightly from that observed in a crystal structure. The most significant difference between the solution and crystal structures occurs in the putative DNA-binding helix-turn-helix (HTH) motif. This is the region of lowest stability in solution and a point of protein-protein contact in the crystal. Alternative conformations of the HTH motif may permit adjustment of the structure for optimal DNA binding.
- Biomolecular Nuclear Magnetic Resonance Laboratory, Department of Chemistry, Graduate Program in Biotechnology Science and Engineering, University of Alabama in Huntsville, Huntsville, Alabama 35899, USA.
Organizational Affiliation: