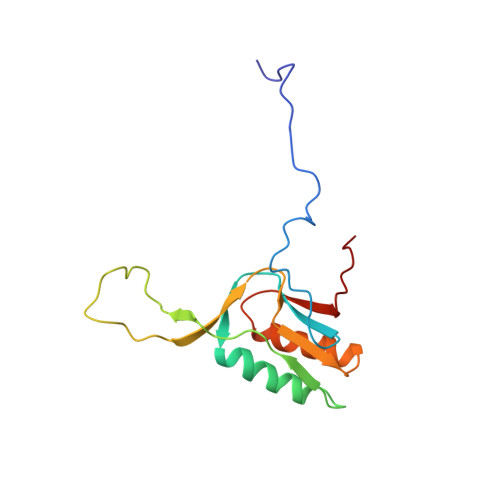
Solution Structure of Ribosomal Protein L16 from Thermus thermophilus HB8
Nishimura, M., Yoshida, T., Shirouzu, M., Terada, T., Kuramitsu, S., Yokoyama, S., Ohkubo, T., Kobayashi, Y.(2004) J Mol Biology 344: 1369-1383
- PubMed: 15561149 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2004.10.011
- Primary Citation Related Structures:
1WKI - PubMed Abstract:
Ribosomal protein L16 is an essential component of the bacterial ribosome. It organizes the architecture of aminoacyl tRNA binding site in the ribosome 50S subunit. The three-dimensional structure of L16 from Thermus thermophilus HB8 was determined by NMR. In solution, L16 forms an alpha+beta sandwich structure combined with two additional beta sheets located at the loop regions connecting the two layers. The terminal regions and a central loop region did not show any specific secondary structure. The structured part of L16 could be superimposed well on the C(alpha) model of L16 determined in the crystal structure of the ribosome 50S subunit. By overlaying the L16 solution structure onto the coordinates of the ribosome crystal structure, we constructed the combined model that represents the ribosome-bound state of L16 in the detailed structure. The model showed that L16 possesses residues in contact with helices 38, 39, 42, 43 and 89 of 23S rRNA and helix 4 of 5S rRNA. This suggests its broad effect on the ribosome architecture. Comparison of L16 with the L10e protein, which is the archaeal counterpart, showed that they share a common fold, but differ in some regions of functional importance, especially in the N-terminal region. All known mutation sites in L16 that confer resistance to avilamycin and evernimicin were positioned so that their side-chains were exposed to solvent in the internal cavity of the ribosome. This suggests the direct participation of L16 as a part of the binding site for antibiotics.
- Graduate School of Pharmaceutical Sciences, Osaka University, 1-6 Yamadaoka, Suita, Osaka 565-0871, Japan.
Organizational Affiliation: