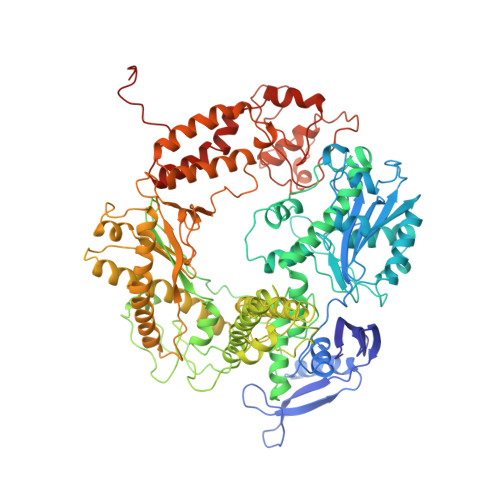
Crystal structure of a pol alpha family replication DNA polymerase from bacteriophage RB69.
Wang, J., Sattar, A.K., Wang, C.C., Karam, J.D., Konigsberg, W.H., Steitz, T.A.(1997) Cell 89: 1087-1099
- PubMed: 9215631
- DOI: https://doi.org/10.1016/s0092-8674(00)80296-2
- Primary Citation Related Structures:
1WAF, 1WAJ - PubMed Abstract:
The 2.8 A resolution crystal structure of the bacteriophage RB69 gp43, a member of the eukaryotic pol alpha family of replicative DNA polymerases, shares some similarities with other polymerases but shows many differences. Although its palm domain has the same topology as other polymerases, except rat DNA polymerase beta, one of the three carboxylates required for nucleotidyl transfer is located on a different beta strand. The structures of the fingers and thumb domains are unrelated to all other known polymerase structures. The editing 3'-5' exonuclease domain of gp43 is homologous to that of E. coli DNA polymerase I but lies on the opposite side of the polymerase active site. An extended structure-based alignment of eukaryotic DNA polymerase sequences provides structural insights that should be applicable to most eukaryotic DNA polymerases.
- Department of Molecular Biophysics and Biochemistry, Yale University, New Haven, Connecticut 06520-8114, USA.
Organizational Affiliation: