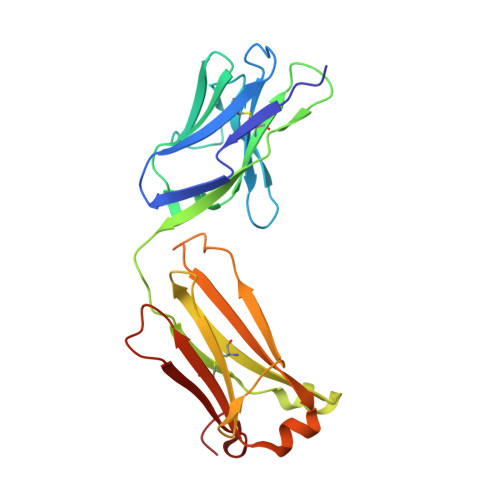
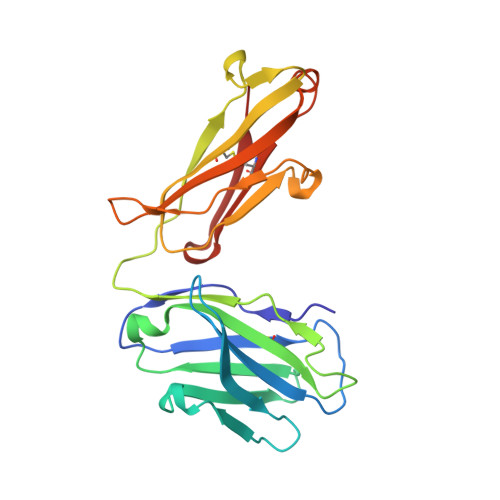
Structural Studies of an Efficient Catalytic Antibody Operating by Ping-Pong and Induced Fit Mechanisms
Beuscher IV, A.E., Reuter, J., Olson, A.J., Romesberg, F.E., Schultz, P.G., Wirsching, P., Janda, K.D., Lerner, R.A., Stevens, R.C.To be published.