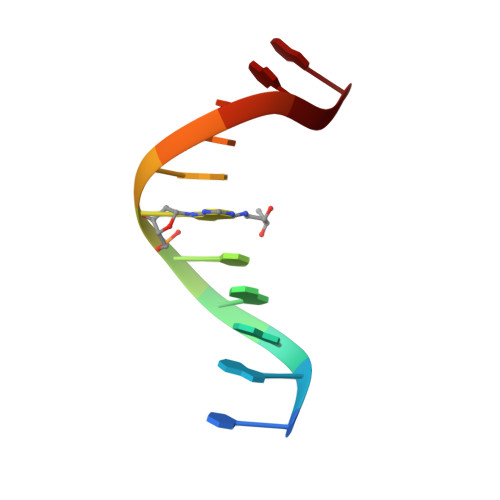
Structure of a site specific major groove (2S,3S)-N6-(2,3,4-trihydroxybutyl)-2'-deoxyadenosyl DNA adduct of butadiene diol epoxide.
Scholdberg, T.A., Nechev, L.V., Merritt, W.K., Harris, T.M., Harris, C.M., Lloyd, R.S., Stone, M.P.(2004) Chem Res Toxicol 17: 717-730
- PubMed: 15206893 Search on PubMed
- DOI: https://doi.org/10.1021/tx034271+
- Primary Citation Related Structures:
1U6C - PubMed Abstract:
The solution structure of the (2S,3S)-N(6)-(2,3,4-trihydroxybutyl)-2'-deoxyadenosyl adduct arising from the alkylation of adenine N(6) at position X(6) in d(CGGACXAGAAG).d(CTTCTTGTCCG), by butadiene diol epoxide, was determined. This oligodeoxynucleotide contains codon 61 (underlined) of the human N-ras protooncogene. This oligodeoxynucleotide, containing the adenine N(6) adduct butadiene triol (BDT) adduct at the second position of codon 61, was named the ras61 S,S-BDT-(61,2) adduct. NMR spectroscopy revealed modest structural perturbations localized to the site of adduction at X(6).T(17), and its nearest-neighbor base pairs C(5).G(18) and A(7).T(16). All sequential NOE connectivities arising from DNA protons were observed. Torsion angle analysis from COSY data suggested that the deoxyribose sugar at X(6) remained in the C2'-endo conformation. Molecular dynamics calculations using a simulated annealing protocol restrained by a total of 442 NOE-derived distances and J coupling-derived torsion angles refined structures in which the BDT moiety oriented in the major groove. Relaxation matrix analysis suggested hydrogen bonding between the hydroxyl group located at the beta-carbon of the BDT moiety and the T(17) O(4) of the modified base pair X(6).T(17). The minimal perturbation of DNA induced by this major groove adduct correlated with its facile bypass by three Escherichia coli DNA polymerases in vitro and its weak mutagenicity [Carmical, J. R., Nechev, L. V., Harris, C. M., Harris, T. M., and Lloyd, R. S. (2000) Environ. Mol. Mutagen. 35, 48-56]. Overall, the structure of this adduct is consistent with an emerging pattern in which major groove adenine N(6) alkylation products of styrene and butadiene oxides that do not strongly perturb DNA structure are not strongly mutagenic.
- Department of Chemistry, Center in Molecular Toxicology, Vanderbilt-Ingram Cancer Center, Vanderbilt University, Nashville, Tennessee 37235, USA.
Organizational Affiliation: