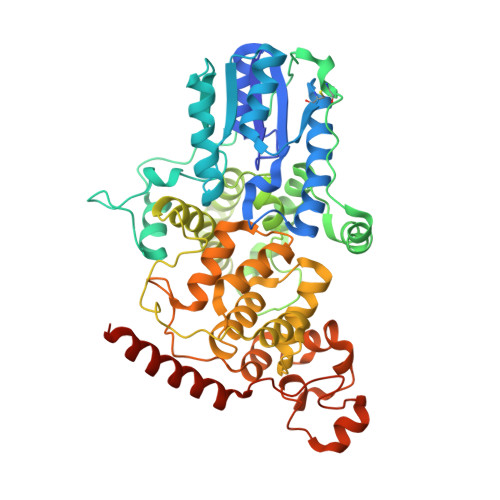
Structure of the photolyase-like domain of cryptochrome 1 from Arabidopsis thaliana.
Brautigam, C.A., Smith, B.S., Ma, Z., Palnitkar, M., Tomchick, D.R., Machius, M., Deisenhofer, J.(2004) Proc Natl Acad Sci U S A 101: 12142-12147
- PubMed: 15299148 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0404851101
- Primary Citation Related Structures:
1U3C, 1U3D - PubMed Abstract:
Signals generated by cryptochrome (CRY) blue-light photoreceptors are responsible for a variety of developmental and circadian responses in plants. The CRYs are also identified as circadian blue-light photoreceptors in Drosophila and components of the mammalian circadian clock. These flavoproteins all have an N-terminal domain that is similar to photolyase, and most have an additional C-terminal domain of variable length. We present here the crystal structure of the photolyase-like domain of CRY-1 from Arabidopsis thaliana. The structure reveals a fold that is very similar to photolyase, with a single molecule of FAD noncovalently bound to the protein. The surface features of the protein and the dissimilarity of a surface cavity to that of photolyase account for its lack of DNA-repair activity. Previous in vitro experiments established that the photolyase-like domain of CRY-1 can bind Mg.ATP, and we observe a single molecule of an ATP analog bound in the aforementioned surface cavity, near the bound FAD cofactor. The structure has implications for the signaling mechanism of CRY blue-light photoreceptors.
- Department of Biochemistry, University of Texas Southwestern Medical Center, 5323 Harry Hines Boulevard, Dallas, TX 75390-9050, USA.
Organizational Affiliation: