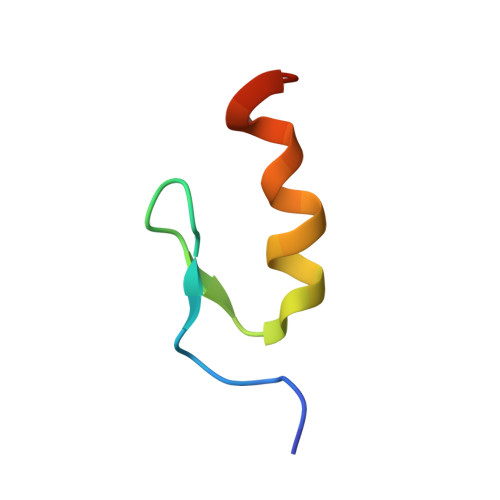
A Classic Zinc Finger from Friend of GATA Mediates an Interaction with the Coiled-coil of Transforming Acidic Coiled-coil 3.
Simpson, R.J.Y., Lee, S.H.Y., Bartle, N., Sum, E.Y., Visvader, J.E., Matthews, J.M., Mackay, J.P., Crossley, M.(2004) J Biological Chem 279: 39789-39797
- PubMed: 15234987 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M404130200
- Primary Citation Related Structures:
1SRK - PubMed Abstract:
Classic zinc finger domains (cZFs) consist of a beta-hairpin followed by an alpha-helix. They are among the most abundant of all protein domains and are often found in tandem arrays in DNA-binding proteins, with each finger contributing an alpha-helix to effect sequence-specific DNA recognition. Lone cZFs, not found in tandem arrays, have been postulated to function in protein interactions. We have studied the transcriptional co-regulator Friend of GATA (FOG), which contains nine zinc fingers. We have discovered that the third cZF of FOG contacts a coiled-coil domain in the centrosomal protein transforming acidic coiled-coil 3 (TACC3). Although FOG-ZF3 exhibited low solubility, we have used a combination of mutational mapping and protein engineering to generate a derivative that was suitable for in vitro and structural analysis. We report that the alpha-helix of FOG-ZF3 recognizes a C-terminal portion of the TACC3 coiled-coil. Remarkably, the alpha-helical surface utilized by FOG-ZF3 is the same surface responsible for the well established sequence-specific DNA-binding properties of many other cZFs. Our data demonstrate the versatility of cZFs and have implications for the analysis of many as yet uncharacterized cZF proteins.
- School of Molecular and Microbial Biosciences, G08, University of Sydney, NSW 2006, Australia.
Organizational Affiliation: