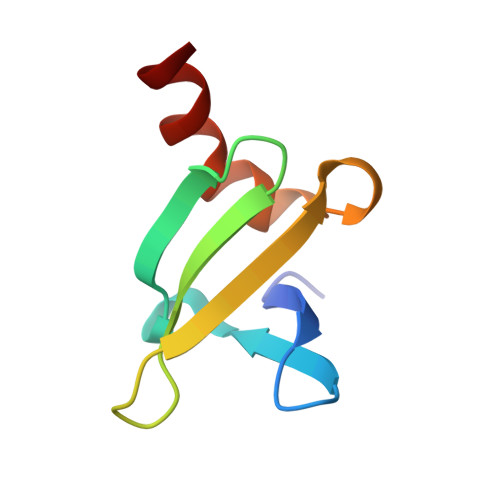
Solution structure of the DNA-binding protein Sac7d from the hyperthermophile Sulfolobus acidocaldarius.
Edmondson, S.P., Qiu, L., Shriver, J.W.(1995) Biochemistry 34: 13289-13304
- PubMed: 7577913 Search on PubMed
- DOI: https://doi.org/10.1021/bi00041a004
- Primary Citation Related Structures:
1SAP - PubMed Abstract:
The Sac7 proteins from the hyperthermophile Sulfolobus acidocaldarius are a heterogeneous mixture of small, thermostable, nonspecific DNA-binding proteins. One of these proteins, Sac7d, has been overexpressed in Escherichia coli to provide a homogeneous preparation for structure, stability, and function studies. We present here essentially complete sequence-specific 1H NMR assignments for Sac7d, a delineation of secondary structural elements, and the high-resolution solution structure obtained from a full relaxation matrix refinement. The final structure provides an excellent fit to the NMR data with an NOE R-factor of 0.27 for backbone NOEs. The structure has a compact globular fold with 82% of the sequence involved in regular secondary structure: an antiparallel two-stranded beta-ribbon with a tight turn, followed by a short 3(10) helix, an antiparallel three-stranded beta-sheet, another short 3(10) helix, and finally four turns of alpha-helix. The amphipathic alpha-helix packs across the hydrophobic face of the three-stranded beta-sheet in an open-faced sandwich arrangement with at least one turn of the helix exposed beyond the sheet. The hydrophobic face of the beta-ribbon packs against a corner of the twisted beta-sheet. The single tryptophan responsible for the 88% fluorescence quenching upon DNA binding is exposed on the surface of the three-stranded beta-sheet. Lysines 5 and 7, whose monomethylation may be associated with enhanced thermostability, are highly solvent exposed along the inner edge of the two-stranded ribbon. The structure of Sac7d differs in many respects from that reported for the homologous native Sso7d [Baumann et al. (1994) Nature Struct. Biol. 1, 808] with a backbone RMSD greater than 3.0 A, largely due to the packing and length of the C-terminal alpha-helix which may be important in Sac7d DNA binding.
- Department of Medical Biochemistry, School of Medicine, Southern Illinois University, Carbondale 62901, USA.
Organizational Affiliation: