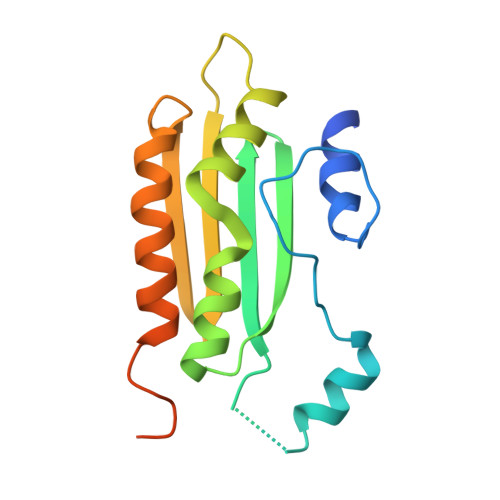
Size selective recognition of siRNA by an RNA silencing suppressor
Vargason, J.M., Szittya, G., Burgyan, J., Hall, T.M.T.(2003) Cell 115: 799-811
- PubMed: 14697199 Search on PubMed
- DOI: https://doi.org/10.1016/s0092-8674(03)00984-x
- Primary Citation Related Structures:
1RPU - PubMed Abstract:
RNA silencing in plants likely exists as a defense mechanism against molecular parasites such as RNA viruses, retrotransposons, and transgenes. As a result, many plant viruses have adapted mechanisms to evade and suppress gene silencing. Tombusviruses express a 19 kDa protein (p19), which has been shown to suppress RNA silencing in vivo and bind silencing-generated and synthetic small interfering RNAs (siRNAs) in vitro. Here we report the 2.5 A crystal structure of p19 from the Carnation Italian ringspot virus (CIRV) bound to a 21 nt siRNA and demonstrate in biochemical and in vivo assays that CIRV p19 protein acts as a molecular caliper to specifically select siRNAs based on the length of the duplex region of the RNA.
- Laboratory of Structural Biology, National Institute of Environmental Health Sciences, National Institutes of Health, Research Triangle Park, NC 27709, USA.
Organizational Affiliation: