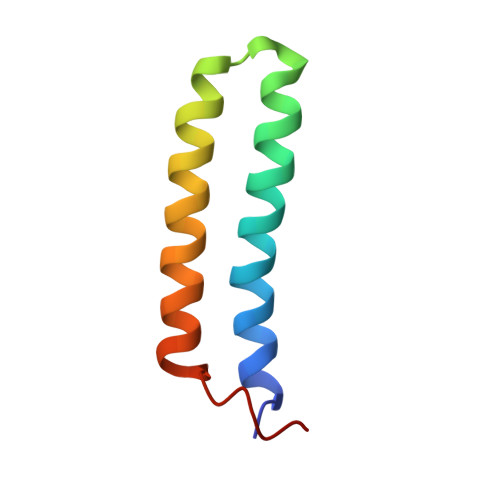
The structure of ColE1 rop in solution.
Eberle, W., Pastore, A., Sander, C., Rosch, P.(1991) J Biomol NMR 1: 71-82
- PubMed: 1841691 Search on PubMed
- DOI: https://doi.org/10.1007/BF01874570
- Primary Citation Related Structures:
1RPR - PubMed Abstract:
The structure of the ColE1 repressor of primer (rop) protein in solution was determined from the proton nuclear magnetic resonance data by a combined use of distance geometry and restrained molecular dynamics calculations. A set of structures was determined with low internal energy and virtually no violations of the experimental distance restraints. Rop forms homodimers: Two helical hairpins are arranged as an antiparallel four helix bundle with a left-handed rope-like twist of the helix axes and with left-handed bundle topology. The very compact packing of the side chains in the helix interfaces of the rop coiled-coil structure may well account for its high stability. Overall, the solution structure is highly similar to the recently determined X-ray structure (Banner, D.W., Kokkinidis, M. and Tsernoglou, D. (1987) J. Mol. Biol., 196, 657-675), although there are minor differences in regions where packing forces appear to influence the crystal structure.
- European Molecular Biology Laboratory, Heidelberg, Germany.
Organizational Affiliation: