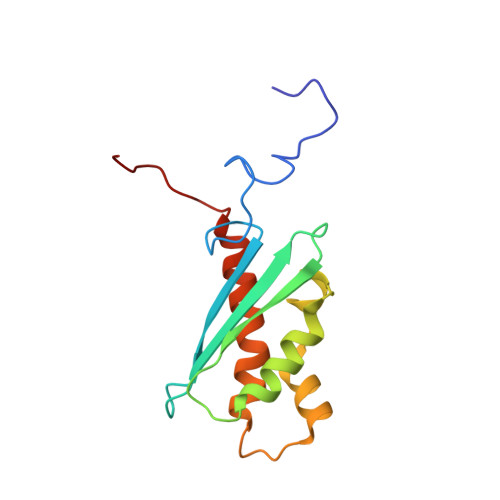
Solution NMR structure of the iron-sulfur cluster assembly protein U (IscU) with zinc bound at the active site.
Ramelot, T.A., Cort, J.R., Goldsmith-Fischman, S., Kornhaber, G.J., Xiao, R., Shastry, R., Acton, T.B., Honig, B., Montelione, G.T., Kennedy, M.A.(2004) J Mol Biology 344: 567-583
- PubMed: 15522305 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2004.08.038
- Primary Citation Related Structures:
1Q48, 1R9P - PubMed Abstract:
IscU is a highly conserved protein that serves as the scaffold for IscS-mediated assembly of iron-sulfur ([Fe-S]) clusters. We report the NMR solution structure of monomeric Haemophilus influenzae IscU with zinc bound at the [Fe-S] cluster assembly site. The compact core of the globular structure has an alpha-beta sandwich architecture with a three-stranded antiparallel beta-sheet and four alpha-helices. A nascent helix is located N-terminal to the core structure. The zinc is ligated by three cysteine residues and one histidine residue that are located in and near conformationally dynamic loops at one end of the IscU structure. Removal of the zinc metal by chelation results in widespread loss of structure in the apo form. The zinc-bound IscU may be a good model for iron-loaded IscU and may demonstrate structural features found in the [Fe-S] cluster bound form. Structural and functional similarities, genomic context in operons containing other homologous genes, and distributions of conserved surface residues support the hypothesis that IscU protein domains are homologous (i.e. derived from a common ancestor) with the SufE/YgdK family of [Fe-S] cluster assembly proteins.
- Biological Sciences Division, Pacific Northwest National Laboratory, Richland, WA 99352, USA.
Organizational Affiliation: