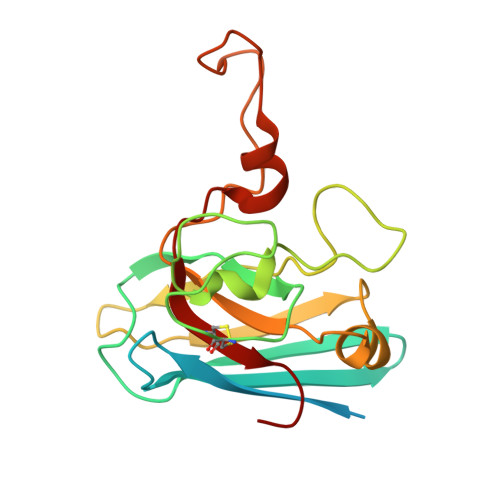
Unique Features of the sodC-encoded Superoxide Dismutase from Mycobacterium tuberculosis, a Fully Functional Copper-containing Enzyme Lacking Zinc in the Active Site.
Spagnolo, L., Toro, I., D'Orazio, M., O'Neill, P., Pedersen, J.Z., Carugo, O., Rotilio, G., Battistoni, A., Djinovic-Carugo, K.(2004) J Biological Chem 279: 33447-33455
- PubMed: 15155722 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M404699200
- Primary Citation Related Structures:
1PZS - PubMed Abstract:
The sodC-encoded Mycobacterium tuberculosis superoxide dismutase (SOD) shows high sequence homology to other members of the copper/zinc-containing SOD family. Its three-dimensional structure is reported here, solved by x-ray crystallography at 1.63-A resolution. Metal analyses of the recombinant protein indicate that the native form of the enzyme lacks the zinc ion, which has a very important structural and functional role in all other known enzymes of this class. The absence of zinc within the active site is due to significant rearrangements in the zinc subloop, including deletion or mutation of the metal ligands His115 and His123. Nonetheless, the enzyme has a catalytic rate close to the diffusion limit; and unlike all other copper/zinc-containing SODs devoid of zinc, the geometry of the copper site is pH-independent. The protein shows a novel dimer interface characterized by a long and rigid loop, which confers structural stability to the enzyme. As the survival of bacterial pathogens within their host critically depends on their ability to recruit zinc in highly competitive environments, we propose that the observed structural rearrangements are required to build up a zinc-independent but fully active and stable copper-containing SOD.
- Structural Biology Laboratory, ELETTRA, Sincrotrone Trieste, Italy.
Organizational Affiliation: