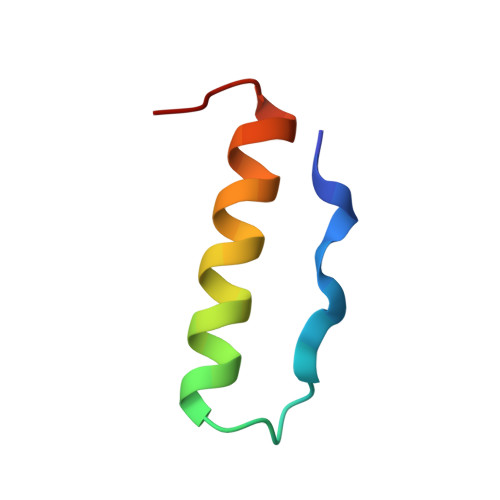
X-ray analysis (1. 4-A resolution) of avian pancreatic polypeptide: Small globular protein hormone.
Blundell, T.L., Pitts, J.E., Tickle, I.J., Wood, S.P., Wu, C.W.(1981) Proc Natl Acad Sci U S A 78: 4175-4179
- PubMed: 16593056 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.78.7.4175
- Primary Citation Related Structures:
1PPT - PubMed Abstract:
The crystal structure of avian pancreatic polypeptide (aPP), a 36-residue polypeptide with some hormonal properties, has been determined by using single isomorphous replacement and anomalous scattering to 2.1-A resolution. The phases were extended to 1.4-A resolution by using a modified tangent formula. The molecule contains two regions of secondary structure-an extended polyproline-like helix (residues 1-8) and an alpha-helix (residues 14-31)-that run roughly antiparallel. The packing together of nonpolar groups from these regions gives the molecule a hydrophobic core in spite of its small size. The aPP molecules form a symmetrical dimer in the crystal stabilized principally by interlocking of nonpolar groups from the alpha-helices. The aPP dimers are crosslinked by coordination of Zn(2+); three aPP molecules contribute ligands to each zinc. The coordination geometry is a distorted trigonal bipyramid. The properties of the aPP molecule in solution are consistent with expectations based on the crystal structure. The aPP molecule has several general features in common with the pancreatic hormones insulin and glucagon. All three hormones have complex mechanisms for self-association. Like insulin, aPP seems to have a stable monomeric structure but its biological activity seems to depend on the more flexible COOH-terminal region analogous to the flexible NH(2)-terminal region of glucagon.
- Laboratory of Molecular Biology, Department of Crystallography, Birkbeck College, University of London, Malet Street, London WC1E 7HX England.
Organizational Affiliation: