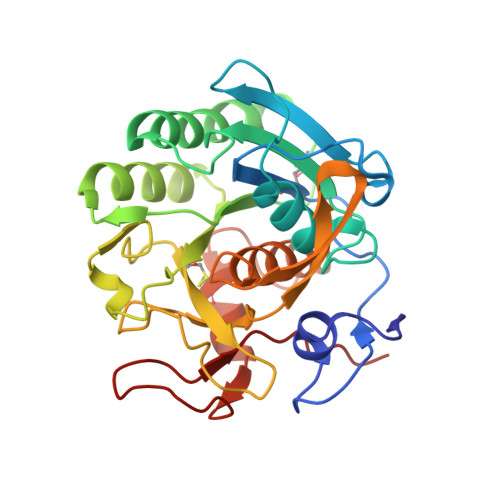
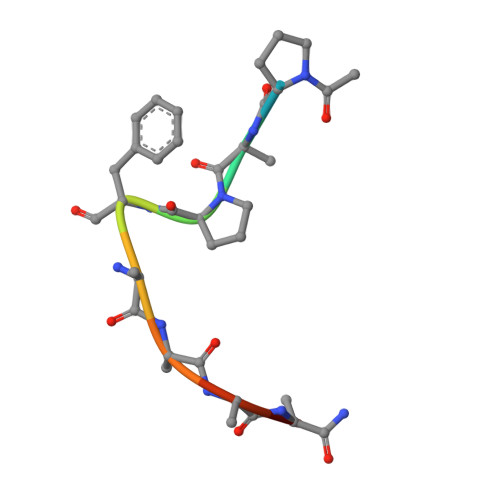
Strategy to design peptide inhibitors: structure of a complex of proteinase K with a designed octapeptide inhibitor N-Ac-Pro-Ala-Pro-Phe-DAla-Ala-Ala-Ala-NH2 at 2.5 A resolution.
Saxena, A.K., Singh, T.P., Peters, K., Fittkau, S., Betzel, C.(1996) Protein Sci 5: 2453-2458
- PubMed: 8976553 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/pro.5560051207
- Primary Citation Related Structures:
1PFG - PubMed Abstract:
The crystal structure of a complex formed by the interaction between proteinase K and a designed octapeptide amide, N-Ac-Pro-Ala-Pro-Phe-DAla-Ala-Ala-Ala-NH2, has been determined at 2.5 A resolution and refined to an R-factor of 16.7% for 7,430 reflections in the resolution range of 8.0-2.50 A. The inhibitor forms a stable complex through a series of hydrogen bonds and hydrophobic interactions with the protein atoms and water molecules. The inhibitor is hydrolyzed between Phe4I and DAla5I (I indicates the inhibitor). The two fragments are separated by a distance of 3.2 A between the carbonyl carbon of Phe4I and the main-chain nitrogen of DAla5I. The N-terminal tetrapeptide occupies subsites S1-S5 (S5 for acetyl group), whereas the C-terminal part fits into S1'-S5' region (S5' for amide group). It is the first time that such an extended electron density for a designed synthetic peptide inhibitor has been observed in the prime region of an enzyme of the subtilisin family. In fact, the inhibitor fills the recognition site completely. There is only a slight rearrangement of the protein residues to accommodate the inhibitor. Superposition of the present octapeptide inhibitor on the hexapeptide inhibitor studied previously shows an overall homology of the two inhibitors, although the individual atoms are displaced significantly. It suggests the existence of a recognition site with flexible dimensions. Kinetic studies indicate an inhibition rate of 100% by this specifically designed peptide inhibitor.
- Department of Biophysics, All India Institute of Medical Sciences, New Delhi, India.
Organizational Affiliation: