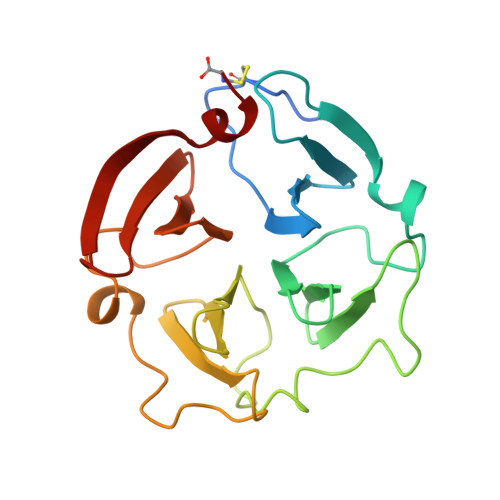
The helping hand of collagenase-3 (MMP-13): 2.7 A crystal structure of its C-terminal haemopexin-like domain.
Gomis-Ruth, F.X., Gohlke, U., Betz, M., Knauper, V., Murphy, G., Lopez-Otin, C., Bode, W.(1996) J Mol Biology 264: 556-566
- PubMed: 8969305 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1996.0661
- Primary Citation Related Structures:
1PEX - PubMed Abstract:
Collagenase-3 (MMP-13) is a matrix metalloproteinase involved in human breast cancer pathology and in arthritic processes. The crystal structure of its C-terminal haemopexin-like domain has been solved by molecular replacement and refined to an R-value of 0.195 using data to 2.7 A resolution. This structure reveals a disk-like shape. The chain is folded into a beta-propeller structure of pseudo 4-fold symmetry, with the four propeller blades arranged around a funnel-like tunnel. This central tunnel tube harbours four ions assigned as two calcium and two chloride ions. The C-terminal domain of collagenase-3 has a similar structure to the equivalent domain of gelatinase A and fibroblast collagenase 1; however, its detailed structure and surface charge pattern has a somewhat greater similarity to the latter, in agreement with the subgrouping of MMP-13 with the collagenase subfamily of MMPs. It is proposed that several small structural differences may act together to confer the characteristic binding and cleavage specificities of collagenases for triple-helical substrates, probably in co-operation with a fitting interdomain linker.
- Max-Planck-Institut für Biochemie, Abteilung für Strukturforschung, Planegg-Martinsreid, Germany.
Organizational Affiliation: