Structure of a hypothetical protein from M. thermautotrophicus reveals a novel fold and a pseudo 2-fold axis of symmetry
Cuff, M.E., Skarina, T., Savchenko, A., Edwards, A., Joachimiak, A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| hypothetical protein | 125 | Methanothermobacter thermautotrophicus str. Delta H | Mutation(s): 0 | 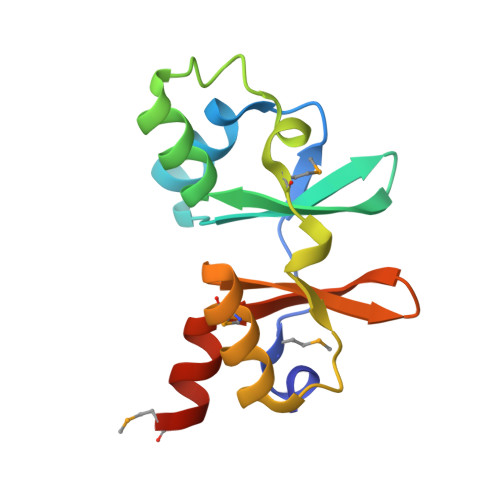 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O27659 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| MG Download:Ideal Coordinates CCD File | B [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 58.788 | α = 90 |
| b = 48.586 | β = 107.78 |
| c = 56.481 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CNS | refinement |
| d*TREK | data reduction |
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |
| SHARP | phasing |