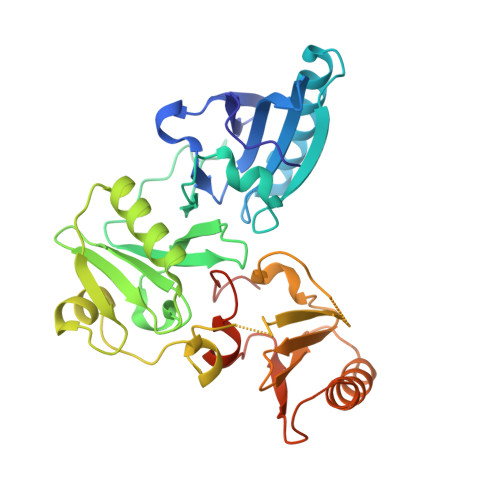
Activation in isolation: Exposure of the actin-binding site in the C-terminal half of gelsolin does not require actin
Narayan, K., Chumnarnsilpa, S., Choe, H., Irobi, E., Urosev, D., Lindberg, U., Schutt, C.E., Burtnick, L.D., Robinson, R.C.(2003) FEBS Lett 552: 82-85
- PubMed: 14527664 Search on PubMed
- DOI: https://doi.org/10.1016/s0014-5793(03)00933-5
- Primary Citation Related Structures:
1P8X - PubMed Abstract:
Gelsolin requires activation to carry out its severing and capping activities on F-actin. Here, we present the structure of the isolated C-terminal half of gelsolin (G4-G6) at 2.0 A resolution in the presence of Ca(2+) ions. This structure completes a triptych of the states of activation of G4-G6 that illuminates its role in the function of gelsolin. Activated G4-G6 displays an open conformation, with the actin-binding site on G4 fully exposed and all three type-2 Ca(2+) sites occupied. Neither actin nor the type-l Ca(2+), which normally is sandwiched between actin and G4, is required to achieve this conformation.
- Department of Medical Biochemistry and Microbiology, Uppsala University, Box 582, 751 23 Uppsala, Sweden.
Organizational Affiliation: