A 1.7A structure of Fve, a member of the new fungal immunomodulatory protein family
Paaventhan, P., Joseph, J.S., Seow, S.V., Vaday, S., Robinson, H., Chua, K.Y., Kolatkar, P.R.(2003) J Mol Biology 332: 461-470
- PubMed: 12948495
- DOI: https://doi.org/10.1016/s0022-2836(03)00923-9
- Primary Citation Related Structures:
1OSY - PubMed Abstract:
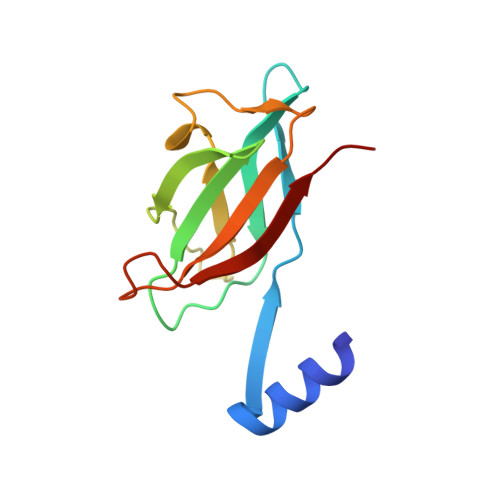
Fve, a major fruiting body protein from Flammulina velutipes, a mushroom possessing immunomodulatory activity, stimulates lymphocyte mitogenesis, suppresses systemic anaphylaxis reactions and edema, enhances transcription of IL-2, IFN-gamma and TNF-alpha, and hemagglutinates red blood cells. It appears to be a lectin with specificity for complex cell-surface carbohydrates. Fve is a non-covalently linked homodimer containing no Cys, His or Met residues. It shares sequence similarity only to the other fungal immunomodulatory proteins (FIPs) LZ-8, Gts, Vvo and Vvl, all of unknown structure. The 1.7A structure of Fve solved by single anomalous diffraction of NaBr-soaked crystals is novel: each monomer consists of an N-terminal alpha-helix followed by a fibronectin III (FNIII) fold. The FNIII fold is the first instance of "pseudo-h-type" topology, a transition between the seven beta-stranded s-type and the eight beta-stranded h-type topologies. The structure suggests that dimerization, critical for the activity of FIPs, occurs by 3-D domain swapping of the N-terminal helices and is stabilized predominantly by hydrophobic interactions. The structure of Fve is the first in this lectin family to be reported, and the first of an FNIII domain-containing protein of fungal origin.
- Genome Institute of Singapore, 117528 Singapore, Singapore.
Organizational Affiliation: