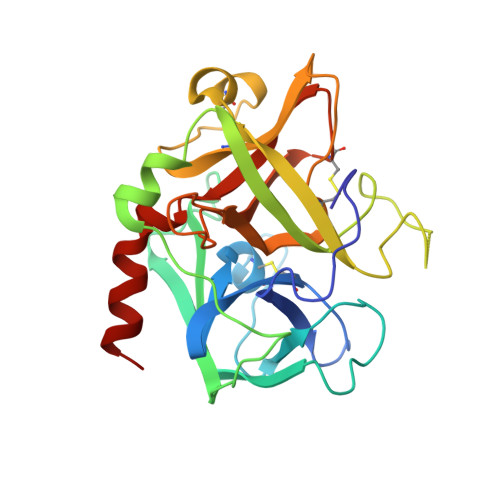
Crystallographic structures of thrombin complexed with thrombin receptor peptides: existence of expected and novel binding modes.
Mathews, I.I., Padmanabhan, K.P., Ganesh, V., Tulinsky, A., Ishii, M., Chen, J., Turck, C.W., Coughlin, S.R., Fenton 2nd., J.W.(1994) Biochemistry 33: 3266-3279
- PubMed: 8136362 Search on PubMed
- DOI: https://doi.org/10.1021/bi00177a018
- Primary Citation Related Structures:
1NRN, 1NRO, 1NRP, 1NRQ, 1NRR, 1NRS - PubMed Abstract:
Many of the vital actions of thrombin on platelets and other cells appear to be mediated by the recently cloned seven-transmembrane-domain thrombin receptor. Thrombin activates this receptor by a novel proteolytic mechanism. The amino-terminal exodomain of the receptor contains the sequence LDPRSFLLRNPNDKYEPF. Structure-activity studies with mutant receptors and receptor peptides suggest that this sequence binds to thrombin at two sites: LDPR with the active center of thrombin and KYEPF with the fibrinogen recognition exosite of thrombin. Thrombin then cleaves the Arg41-Ser42 bond to unmask a new amino terminus, which functions as a tethered peptide ligand binding to as yet undefined sites within the body of the receptor to effect receptor activation. We have determined eight crystal structures of thrombin complexed with receptor-based peptides. Each of the two components of the bidentate docking model was captured in individual cocrystals. In one crystal type, the LDPR sequence docked in the active center of thrombin in a manner analogous to d-PheProArg chloromethyl ketone. In other crystals, the KYEPF sequence bound in the fibrinogen anion binding exosite of thrombin in a manner analogous to the DFEEI sequence of the carboxylate-terminal peptide of hirudin. Strikingly, however, generation of a single crystal that includes both components of the anticipated bidentate binding mode was not achieved, apparently because the peptides have a dominant solution S-like conformation that does not bind in a productive way at the active center. This peptide structure apparently favored a novel alternative mode of receptor peptide-thrombin interaction in which the receptor peptides formed an intermolecular bridge between neighboring thrombin molecules, resulting in an infinite peptide thrombin chain in crystals. In this structure, the KYEPF sequence docked in the expected manner at the exosite of one thrombin molecule, but the LDPR sequence docked in an unusual nonproductive mode with the active center of a neighboring molecule. Mutations that removed important determinants of the S-like receptor peptide structure underlying the bridging mode in the receptor itself did not significantly alter thrombin signaling. Additionally, a comparison of receptor density to the responsiveness of a cell did not support a role for receptor oligomerization in signaling. The physiological role for this unexpected intermolecular binding mode, if any, remains to be identified.(ABSTRACT TRUNCATED AT 400 WORDS)
- Department of Chemistry, Michigan State University, East Lansing 48824-1322.
Organizational Affiliation: