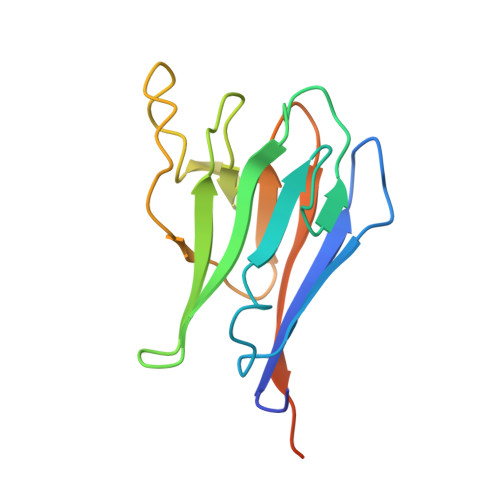
NMR Structure of the forkhead-associated domain from the Arabidopsis receptor kinase-associated protein phosphatase.
Lee, G., Ding, Z., Walker, J.C., Van Doren, S.R.(2003) Proc Natl Acad Sci U S A 100: 11261-11266
- PubMed: 14500786
- DOI: https://doi.org/10.1073/pnas.2031918100
- Primary Citation Related Structures:
1MZK - PubMed Abstract:
Forkhead-associated (FHA) domains are phosphoprotein-binding modules found in diverse signaling proteins that bind partners phosphorylated on threonine or serine. Kinase-associated protein phosphatase from Arabidopsis employs its FHA domain for negative regulation of receptor-like kinase signaling pathways, which are important in plant development. The solution structure of the free state of kinase-interacting FHA domain (KI-FHA) of kinase-associated protein phosphatase has been determined with high precision and accuracy using residual dipolar couplings. KI-FHA is a sandwich of a five-stranded mixed beta-sheet with a six-stranded antiparallel beta-sheet. Despite homology only in the recognition loops, this fold is shared with FHA domains from checkpoint proteins from yeast and humans, as well as with nonhomologous MH2 domains of Smad tumor suppressors. A shared pattern of hydrophobicity throughout FHA domains and Smad MH2 domains may stabilize the core of the beta-sandwich. Evolutionary trace analysis of FHA domains suggests class-specific residues in the recognition loops that could tune their phosphoprotein-binding specificity. This surface agrees with that of KI-FHA in contact with a phosphothreonine peptide ligand. Evolutionary trace analysis also predicts an unexpected swath of class-specific residues on another face of FHA domains. Protein interactions with these faces may affect assembly of transmembrane signaling complexes in plants, and in other FHA domain-containing assemblies.
- Department of Biochemistry, 117 Schweitzer Hall, and Division of Biological Sciences, 105 Tucker Hall, University of Missouri, Columbia, MO 65211, USA.
Organizational Affiliation: