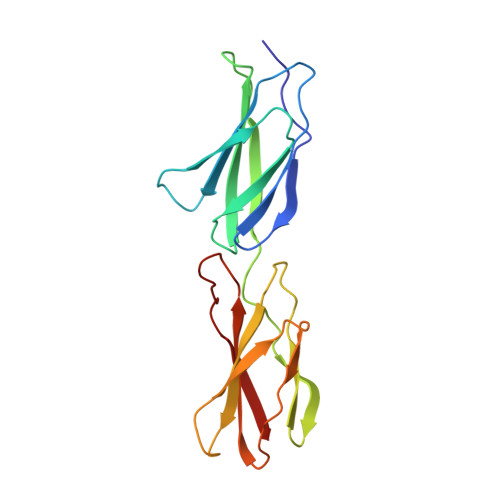
Solution structure and dynamics of linked cell attachment modules of mouse fibronectin containing the RGD and synergy regions: comparison with the human fibronectin crystal structure.
Copie, V., Tomita, Y., Akiyama, S.K., Aota, S., Yamada, K.M., Venable, R.M., Pastor, R.W., Krueger, S., Torchia, D.A.(1998) J Mol Biology 277: 663-682
- PubMed: 9533887 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1998.1616
- Primary Citation Related Structures:
1MFN, 2MFN - PubMed Abstract:
We report the three-dimensional solution structure of the mouse fibronectin cell attachment domain consisting of the linked ninth and tenth type III modules, mFnFn3(9,10). Because the tenth module contains the RGD cell attachment sequence while the ninth contains the synergy region, mFnFn3(9,10) has the cell attachment activity of intact fibronectin. Essentially complete signal assignments and approximately 1800 distance and angle restraints were derived from multidimensional heteronuclear NMR spectra. These restraints were used with a hybrid distance geometry/simulated annealing protocol to generate an ensemble of 20 NMR structures having no distance or angle violations greater than 0.3 A or 3 degrees. Although the beta-sheet core domains of the individual modules are well-ordered structures, having backbone atom rmsd values from the mean structure of 0.51(+/-0.12) and 0.40(+/-0.07) A, respectively, the rmsd of the core atom coordinates increases to 3.63(+/-1.41) A when the core domains of both modules are used to align the coordinates. The latter result is a consequence of the fact that the relative orientation of the two modules is not highly constrained by the NMR restraints. Hence, while structures of the beta-sheet core domains of the NMR structures are very similar to the core domains of the crystal structure of hFnFn3(9,10), the ensemble of NMR structures suggests that the two modules form a less extended and more flexible structure than the fully extended rod-like crystal structure. The radius of gyration, Rg, of mFnFn3(9,10) derived from small-angle neutron scattering measurements, 20.5(+/-0.5) A, agrees with the average Rg calculated for the NMR structures, 20.4 A, and is ca 1 A less than the value of Rg calculated for the X-ray structure. The values of the rotational anisotropy, D ||/D perpendicular, derived from an analysis of 15N relaxation data, range from 1.7 to 2.1, and are significantly less than the anisotropy of 2.67 predicted by hydrodynamic modeling of the crystal coordinates. In contrast, hydrodynamic modeling of the NMR coordinates yields anisotropies in the range of 1.9 to 2.7 (average 2.4(+/-0.2)), with NMR structures bent by more than 20 degrees relative the crystal structure having calculated anisotropies in best agreement with experiment. In addition, the relaxation parameters indicate that several loops in mFnFn3(9,10), including the RGD loop, are flexible on the nanosecond to picosecond time-scale. Taken together, our results suggest that, in solution, the limited set of interactions between the mFnFn3(9,10) modules position the RGD and synergy regions to interact specifically with cell surface integrins, and at the same time permit sufficient flexibility that allows mFnFn3(9,10) to adjust for some variation in integrin structure or environment.
- Molecular Structural Biology Unit, National Institute of Dental Research, National Institutes of Health, Bethesda, MD 20892, USA.
Organizational Affiliation: