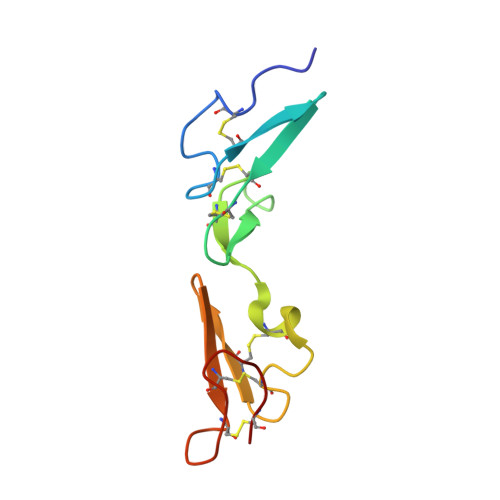
Solution Structure and Dynamics of a Calcium Binding Epidermal Growth Factor-like Domain Pair from the Neonatal Region of Human Fibrillin-1.
Smallridge, R.S., Whiteman, P., Werner, J.M., Campbell, I.D., Handford, P.A., Downing, A.K.(2003) J Biological Chem 278: 12199-12206
- PubMed: 12511552 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M208266200
- Primary Citation Related Structures:
1LMJ - PubMed Abstract:
Fibrillin-1 is a mosaic protein mainly composed of 43 calcium binding epidermal growth factor-like (cbEGF) domains arranged as multiple, tandem repeats. Mutations within the fibrillin-1 gene cause Marfan syndrome (MFS), a heritable disease of connective tissue. More than 60% of MFS-causing mutations identified are localized to cbEGFs, emphasizing that the native properties of these domains are critical for fibrillin-1 function. The cbEGF12-13 domain pair is within the longest run of cbEGFs, and many mutations that cluster in this region are associated with severe, neonatal MFS. The NMR solution structure of Ca(2+)-loaded cbEGF12-13 exhibits a near-linear, rod-like arrangement of domains. This observation supports the hypothesis that all fibrillin-1 (cb)EGF-cbEGF pairs, characterized by a single interdomain linker residue, possess this rod-like structure. The domain arrangement of cbEGF12-13 is stabilized by additional interdomain packing interactions to those observed for cbEGF32-33, which may help to explain the previously reported higher calcium binding affinity of cbEGF13. Based on this structure, a model of cbEGF11-15 that encompasses all known neonatal MFS missense mutations has highlighted a potential binding region. Backbone dynamics data confirm the extended structure of cbEGF12-13 and lend support to the hypothesis that a correlation exists between backbone flexibility and cbEGF domain calcium affinity. These results provide important insight into the potential consequences of MFS-associated mutations for the assembly and biomechanical properties of connective tissue microfibrils.
- Divisions of Structural Biology, Department of Biochemistry, University of Oxford, South Parks Road, Oxford OX1 3QU, United Kingdom.
Organizational Affiliation: