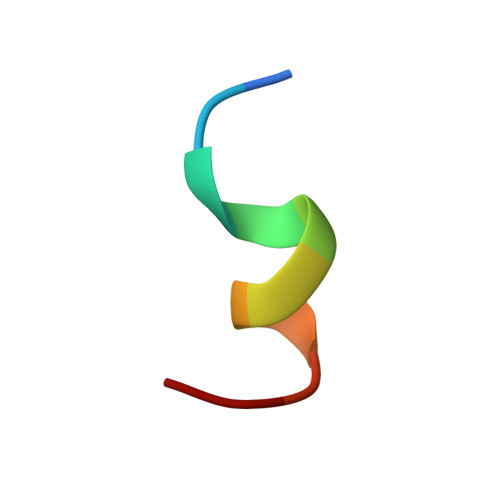Solution structures of the cytoplasmic tail complex from platelet integrin alpha IIb- and beta 3-subunits.
Weljie, A.M., Hwang, P.M., Vogel, H.J.(2002) Proc Natl Acad Sci U S A 99: 5878-5883
- PubMed: 11983888 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.092515799
- Primary Citation Related Structures:
1KUP, 1KUZ - PubMed Abstract:
Integrin adhesion receptors constitute a cell-signaling system whereby interactions in the small cytoplasmic domains of the heterodimeric alpha- and beta-subunits provoke major functional alterations in the large extracellular domains. With two-dimensional NMR spectroscopy, we examined two synthetic peptides [alphaIIb((987)MWKVGFFKRNR) and beta3((716)KLLITIHDRKEFAKFEEERARAKWD)] encompassing the membrane-proximal regions of the cytoplasmic domain motifs from the platelet integrin complex alphaIotaIotabbeta3. These membrane-proximal regions contain two conserved motifs, represented by (989)KVGFFKR in the alphaIIb-subunit, and (716)KLLITIHDR in the beta3-subunit. The dimer interaction consists of two adjacent helices with residues V990 and F993 of the alphaIotaIotab-subunit heavily implicated in the dimer interfacial region, as is I719 of beta3. These residues are situated within the conserved motifs of their respective proteins. Further structural analysis of this unique peptide heterodimer suggests that two distinct conformers are present. The major structural difference between the two conformers is a bend in the beta3-peptide between D723 and A728, whereas the helical character in the other regions remains intact. Earlier mutational analysis has shown that a salt bridge between the side chains of alphaIotaIotab(R955) and beta3(D723) is formed. When this ion pair was modeled into both conformers, increased nuclear Overhauser effect violations suggested that the more bent structure was less able to accommodate this interaction. These results provide a molecular level rationalization for previously reported biochemical studies, as well as a basis for an atomic level understanding of the intermolecular interactions that regulate integrin activity.
- Department of Biological Sciences, University of Calgary, 2500 University Drive NW, Calgary, AB, Canada.
Organizational Affiliation: