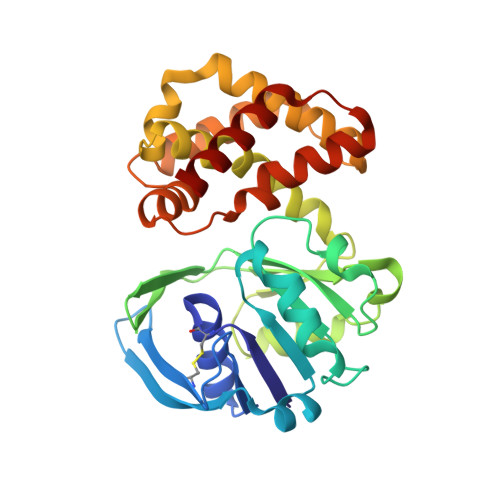
Crystal structure of Escherichia coli ketopantoate reductase at 1.7 A resolution and insight into the enzyme mechanism.
Matak-Vinkovic, D., Vinkovic, M., Saldanha, S.A., Ashurst, J.L., von Delft, F., Inoue, T., Miguel, R.N., Smith, A.G., Blundell, T.L., Abell, C.(2001) Biochemistry 40: 14493-14500
- PubMed: 11724562 Search on PubMed
- DOI: https://doi.org/10.1021/bi011020w
- Primary Citation Related Structures:
1KS9 - PubMed Abstract:
Ketopantoate reductase (KPR, EC 1.1.1.169) catalyzes the NADPH-dependent reduction of ketopantoate to pantoate on the pantothenate (vitamin B(5)) biosynthetic pathway. The Escherichia coli panE gene encoding KPR was cloned and expressed at high levels as the native and selenomethionine-substituted (SeMet) proteins. Both native and SeMet recombinant proteins were purified by three chromatographic steps, to yield pure proteins. The wild-type enzyme was found to have a K(M)(NADPH) of 20 microM, a K(M)(ketopantoate) of 60 microM, and a k(cat) of 40 s(-1). Regular prismatic KPR crystals were prepared using the hanging drop technique. They belonged to the tetragonal space group P4(2)2(1)2, with cell parameters: a = b = 103.7 A and c = 55.7 A, accommodating one enzyme molecule per asymmetric unit. The structure of KPR was determined by the multiwavelength anomalous dispersion method using the SeMet protein, for which data were collected to 2.3 A resolution. The native data were collected to 1.7 A resolution and used to refine the final structure. The secondary structure comprises 12 alpha-helices, three 3(10)-helices, and 11 beta-strands. The enzyme is monomeric and has two domains separated by a cleft. The N-terminal domain has an alphabeta-fold of the Rossmann type. The C-terminal domain (residues 170-291) is composed of eight alpha-helices. KPR is shown to be a member of the 6-phosphogluconate dehydrogenase C-terminal domain-like superfamily. A model for the ternary enzyme-NADPH-ketopantoate ternary complex provides a rationale for kinetic data reported for specific site-directed mutants.
- Department of Biochemistry, University of Cambridge, UK.
Organizational Affiliation: