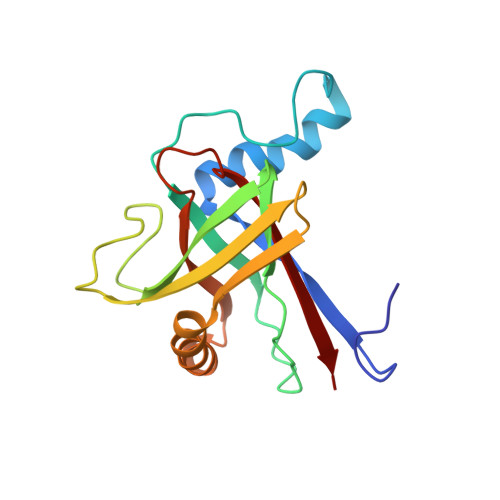
Olfactory marker protein (OMP) exhibits a beta-clam fold in solution: implications for target peptide interaction and olfactory signal transduction.
Baldisseri, D.M., Margolis, J.W., Weber, D.J., Koo, J.H., Margolis, F.L.(2002) J Mol Biology 319: 823-837
- PubMed: 12054873 Search on PubMed
- DOI: https://doi.org/10.1016/S0022-2836(02)00282-6
- Primary Citation Related Structures:
1JYT - PubMed Abstract:
Olfactory marker protein (OMP) is a ubiquitous, cytoplasmic protein found in mature olfactory receptor neurons of all vertebrates. Electrophysiological and behavioral studies demonstrate that it is a modulator of the olfactory signal transduction pathway. Here, we demonstrate that the solution structure of OMP, as determined by NMR studies, is a single globular domain protein comprised of eight beta-strands forming two beta-sheets oriented orthogonally to one another, thus exhibiting a "beta-clam" or "beta-sandwich" fold: beta-sheet 1 is comprised of beta3-beta8-beta1-beta2 and beta-sheet 2 contains beta6-beta5-beta4-beta7. Insertions include two, long alpha-helices located on opposite sides of the beta-clam and three flexible loops. The juxtaposition of beta-strands beta6-beta5-beta4-beta7-beta2-beta1-beta8-beta3 forms a continuously curved surface and encloses one side of the beta-clam. The "cleft" formed by the two beta-sheets is opposite to the closed end of the beta-clam. Using a peptide titration series, we have identified this cleft as the binding surface for a peptide derived from the Bex1 protein. The highly conserved Omega-loop structure adjacent to the Bex1 peptide-binding surface found in OMP may be the site of additional OMP-protein interactions related to its role in modulating olfactory signal transduction. Thus, the interaction between the OMP and Bex1 proteins could facilitate the interaction between OMP and other components of the olfactory signaling pathway.
- Department of Biochemistry and Molecular Biology, University of Maryland School of Medicine, 108 North Greene Street, Baltimore, MD 21201-1503, USA dbaldiss@umaryland.edu
Organizational Affiliation: