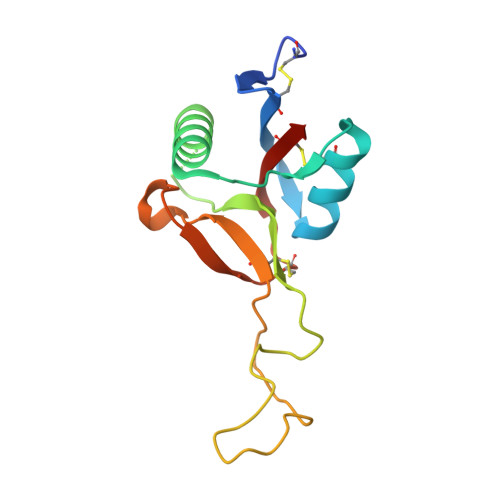
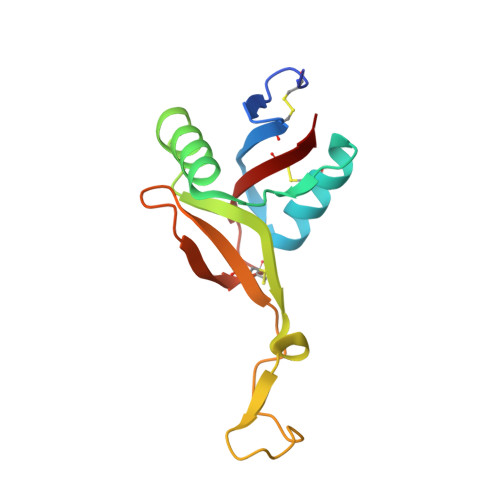
Crystal structure of bitiscetin, a von Willebrand factor-dependent platelet aggregation inducer.
Hirotsu, S., Mizuno, H., Fukuda, K., Qi, M.C., Matsui, T., Hamako, J., Morita, T., Titani, K.(2001) Biochemistry 40: 13592-13597
- PubMed: 11695907 Search on PubMed
- DOI: https://doi.org/10.1021/bi0114933
- Primary Citation Related Structures:
1JWI - PubMed Abstract:
Bitiscetin, a C-type lectin-like protein isolated from the venom of the snake Bitis arientans, promotes the interactions between plasma von Willebrand factor (VWF) and platelet membrane glycoprotein Ib (GPIb) to induce platelet aggregation. We report here the crystal structure of bitiscetin at 2.0 A resolution. The overall fold is similar to those of coagulation factor IX/X-binding protein (IX/X-bp) and flavocetin-A (a GPIb-binding protein), although these three proteins are functionally distinct from one another. The characteristic property determining target recognition is explained mainly by the differences in the surface potential on the central concave surface. A negatively charged patch on the surface of bitiscetin is a candidate for the site of binding to the positively charged surface of the VWF A1 domain, as shown in the case of another platelet aggregation inducer, botrocetin. However, a positively charged patch near the central concave surface is unique for bitiscetin and suggests that it is the binding site for the negatively charged surface of the VWF A3 domain. Thus, the interactions accounting for VWF activation by bitiscetin possibly involve both the A1 and A3 domains of VWF, indicating a specific mechanism of VWF activation by bitiscetin.
- Department of Biochemistry, National Institute of Agrobiological Sciences, Tsukuba, Ibaraki 305-8602, Japan.
Organizational Affiliation: