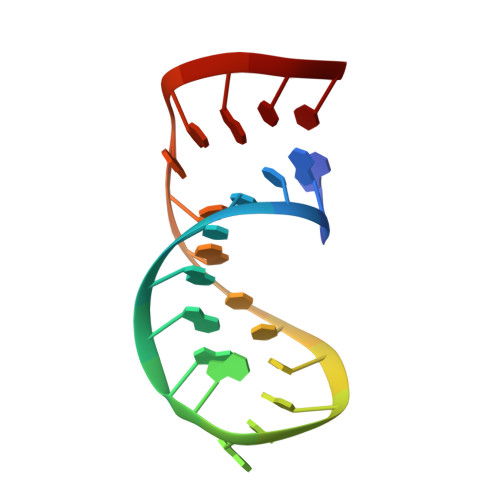
The structure of helix III in Xenopus oocyte 5 S rRNA: an RNA stem containing a two-nucleotide bulge.
Huber, P.W., Rife, J.P., Moore, P.B.(2001) J Mol Biology 312: 823-832
- PubMed: 11575935
- DOI: https://doi.org/10.1006/jmbi.2001.4966
- Primary Citation of Related Structures:
1JUR - PubMed Abstract:
The solution structure of an oligonucleotide containing the helix III sequence from Xenopus oocyte 5 S rRNA has been determined by NMR spectroscopy. Helix III includes two unpaired adenosine residues, flanked on either side by G:C base-pairs, that are required for binding of ribosomal protein L5. The consensus conformation of helix III in the context provided by this oligonucleotide has the two adenosine residues located in the minor groove and stacked upon the 3' flanking guanosine residue, consistent with biochemical studies of free 5 S rRNA in solution. A distinct break in stacking that occurs between the first adenosine residue of the bulge and the flanking 5' guanosine residue exposes the base of the adenosine residue in the minor groove and the base of the guanosine residue in the major groove. The major groove of the helix is widened at the site of the unpaired nucleotides and the helix is substantially bent; nonetheless, the G:C base-pairs flanking the bulge are intact. The data indicate that there may be conformational heterogeneity centered in the bulge region. The corresponding adenosine residues in the Haloarcula marismortui 50 S ribosomal subunit form a dinucleotide platform, which is quite different from the motif seen in solution. Thus, the conformation of helix III probably changes when 5 S rRNA is incorporated into the ribosome.
- Department of Chemistry and Biochemistry, University of Notre Dame, Notre Dame, IN 46556, USA. huber.1@nd.edu
Organizational Affiliation: