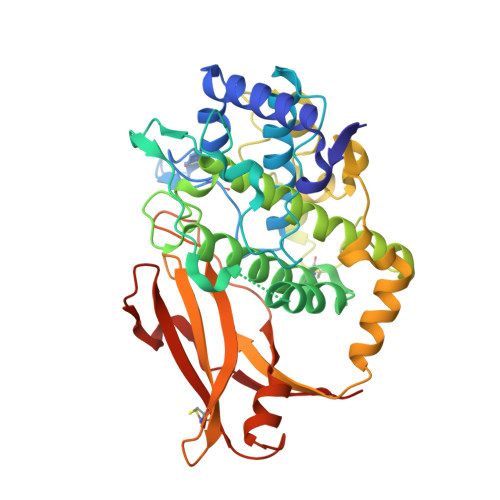
Crystal structure of a functional unit from Octopus hemocyanin.
Cuff, M.E., Miller, K.I., van Holde, K.E., Hendrickson, W.A.(1998) J Mol Biology 278: 855-870
- PubMed: 9614947 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1998.1647
- Primary Citation Related Structures:
1JS8 - PubMed Abstract:
Hemocyanins are giant oxygen transport proteins found in many arthropods and molluscs. Freely dissolved in the hemolymph, they are multisubunit proteins that contain many copies of the active site, a copper atom pair that reversibly binds oxygen. Octopus hemocyanin is composed of ten subunits, each of which contain seven oxygen-binding "functional units". The carboxyl-terminal 47 kDa functional unit, Odg, is a proteolytic isolate that binds oxygen reversibly while exhibiting slight Bohr and magnesium ion effects. In this work we present the X-ray structure determination and analysis of Odg at 2.3 A resolution. Odg has two structural domains: a largely alpha-helical copper binding domain, and a five-stranded anti-parallel beta-sandwich with the jelly roll topology found in many viruses. Six histidine residues ligate the copper atoms, one of which is involved in a thioether bridge. The results show that the hemocyanin from the mollusc and that from the arthropod have distinct tertiary folds in addition to the long recognized differences in their quaternary structures. Nonetheless, a comparison of Octopus and horseshoe crab hemocyanin reveals a similar active site, in a striking example of perhaps both convergent and divergent evolution.
- Department of Biochemistry and Molecular Biophysics, Columbia University, New York, NY 10032, USA.
Organizational Affiliation: