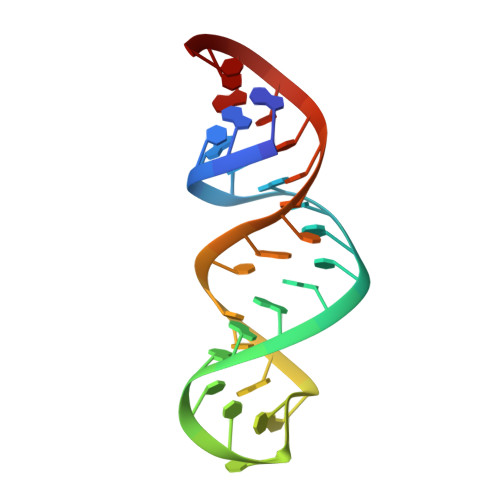
Structural features of an influenza virus promoter and their implications for viral RNA synthesis.
Bae, S.H., Cheong, H.K., Lee, J.H., Cheong, C., Kainosho, M., Choi, B.S.(2001) Proc Natl Acad Sci U S A 98: 10602-10607
- PubMed: 11553808
- DOI: https://doi.org/10.1073/pnas.191268798
- Primary Citation Related Structures:
1JO7 - PubMed Abstract:
The influenza A virus, a severe pandemic pathogen, has a segmented RNA genome consisting of eight single-stranded RNA molecules. The 5' and 3' ends of each RNA segment recognized by the influenza A virus RNA-dependent RNA polymerase direct both transcription and replication of the virus's RNA genome. Promoter binding by the viral RNA polymerase and formation of an active open complex are prerequisites for viral replication and proliferation. Here we describe the solution structure of this promoter as solved by multidimensional, heteronuclear magnetic resonance spectroscopy. Our studies show that the viral promoter has a significant dynamic nature and reveal an unusual displacement of an adenosine that forms a novel (A-A) x U motif and a C-A mismatch stacked in a helix. The characterized structural features of the promoter imply that the specificity of polymerase binding results from an internal RNA loop. In addition, an unexpected bending (46 +/- 10 degrees ) near the initiation site suggests the existence of a promoter recognition mechanism similar to that of DNA-dependent RNA polymerase and a possible regulatory function for the terminal structure during open complex formation.
- Department of Chemistry and National Creative Research Initiative Center, Korea Advanced Institute of Science and Technology, 373-1 Kusung-dong, Yusung-gu, Taejon 305-701, Korea.
Organizational Affiliation: