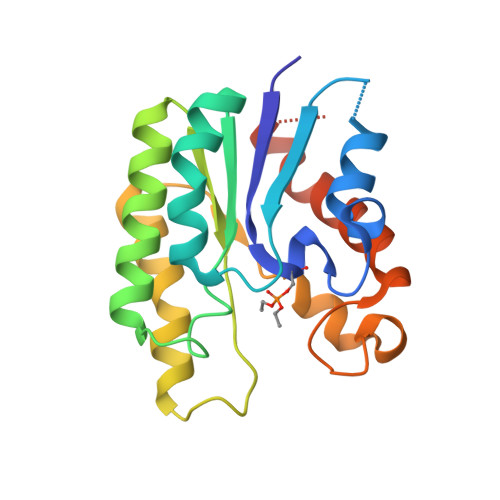
Crystal Structure of Escherichia coli Thioesterase I/Protease I/Lysophospholipase L1: Consensus Sequence Blocks Constitute the Catalytic Center of SGNH-hydrolases through a Conserved Hydrogen Bond Network
Lo, Y.-C., Lin, S.-C., Shaw, J.-F., Liaw, Y.-C.(2003) J Mol Biology 330: 539-551
- PubMed: 12842470 Search on PubMed
- DOI: https://doi.org/10.1016/s0022-2836(03)00637-5
- Primary Citation Related Structures:
1IVN, 1J00, 1JRL - PubMed Abstract:
Escherichia coli thioesterase I (TAP) is a multifunctional enzyme possessing activities of thioesterase, esterase, arylesterase, protease, and lysophospholipase. In particular, TAP has stereoselectivity for amino acid derivative substrates, hence it is useful for the kinetic resolution of racemic mixtures of industrial chemicals. In the present work, the crystal structure of native TAP was determined at 1.9A, revealing a minimal SGNH-hydrolase fold. The structure of TAP in complex with a diethyl phosphono moiety (DEP) identified its catalytic triad, Ser10-Asp154-His157, and oxyanion hole, Ser10-Gly44-Asn73. The oxyanion hole of TAP consists of three residues each separated from the other by more than 3.5A, implying that all of them are highly polarized when substrate bound. The catalytic (His)C(epsilon1)-H...O=C hydrogen bond usually plays a role in the catalytic mechanisms of most serine hydrolases, however, there were none present in SGNH-hydrolases. We propose that the existence of the highly polarized tri-residue-constituted oxyanion hole compensates for the lack of a (His)C(epsilon1)-H...O=C hydrogen bond. This suggests that members of the SGNH-hydrolase family may employ a unique catalytic mechanism. In addition, most SGNH-hydrolases have low sequence identities and presently there is no clear criterion to define consensus sequence blocks. Through comparison of TAP and the three SGNH-hydrolase structures currently known, we have identified a unique hydrogen bond network which stabilizes the catalytic center: a newly discovered structural feature of SGNH-hydrolases. We have defined these consensus sequence blocks providing a basis for the sub-classification of SGNH-hydrolases.
- Graduate Institute of Life Sciences, National Defense Medical Center, Taipei, Neihu 114, Taiwan.
Organizational Affiliation: