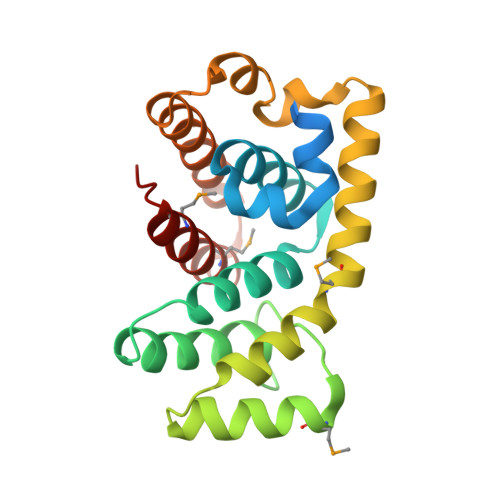
Structure of the RGS-like domain from PDZ-RhoGEF: linking heterotrimeric g protein-coupled signaling to Rho GTPases.
Longenecker, K.L., Lewis, M.E., Chikumi, H., Gutkind, J.S., Derewenda, Z.S.(2001) Structure 9: 559-569
- PubMed: 11470431 Search on PubMed
- DOI: https://doi.org/10.1016/s0969-2126(01)00620-7
- Primary Citation Related Structures:
1HTJ - PubMed Abstract:
The multidomain PDZ-RhoGEF is one of many known guanine nucleotide exchange factors that upregulate Rho GTPases. PDZ-RhoGEF and related family members play a critical role in a molecular signaling pathway from heterotrimeric G protein-coupled receptors to Rho proteins. A approximately 200 residue RGS-like (RGSL) domain in PDZ-RhoGEF and its homologs is responsible for the direct association with Galpha12/13 proteins. To better understand structure-function relationships, we initiated crystallographic studies of the RGSL domain from human PDZ-RhoGEF. A recombinant construct of the RGSL domain was expressed in Escherichia coli and purified, but it did not crystallize. Alternative constructs were designed based on a novel strategy of targeting lysine and glutamic acid residues for mutagenesis to alanine. A triple-point mutant functionally identical to the wild-type protein was crystallized, and its structure was determined by the MAD method using Se-methionine (Se-Met) incorporation. A molecular model of the RGSL domain was refined at 2.2 A resolution, revealing an all-helical tertiary fold with the mutations located at intermolecular lattice contacts. The first nine helices adopt a fold similar to that observed for RGS proteins, although the sequence identity with other such known structures is below 20%. The last three helices are an integral extension of the RGS fold, packing tightly against helices 3 and 4 with multiple hydrophobic interactions. Comparison with RGS proteins suggests features that are likely relevant for interaction with G proteins. Finally, we conclude that the strategy used to produce crystals was beneficial and might be applicable to other proteins resistant to crystallization.
- Department of Molecular Physiology and Biological Physics, University of Virginia, P.O. Box 800736, Charlottesville, VA 22908, USA.
Organizational Affiliation: