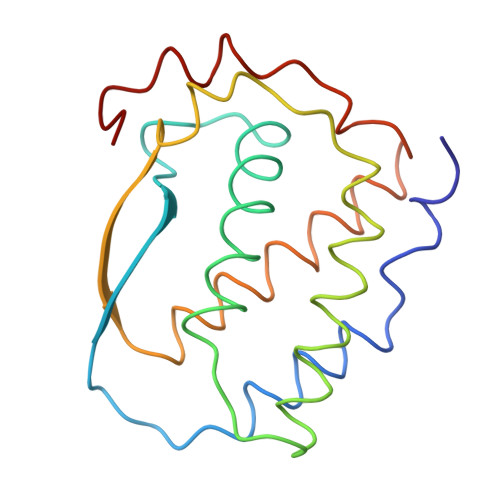
Three-dimensional structure of dimeric human recombinant macrophage colony-stimulating factor.
Pandit, J., Bohm, A., Jancarik, J., Halenbeck, R., Koths, K., Kim, S.H.(1992) Science 258: 1358-1362
- PubMed: 1455231 Search on PubMed
- DOI: https://doi.org/10.1126/science.1455231
- Primary Citation Related Structures:
1HMC - PubMed Abstract:
Macrophage colony-stimulating factor (M-CSF) triggers the development of cells of the monocyte-macrophage lineage and has a variety of stimulatory effects on mature cells of this class. The biologically active form of M-CSF is a disulfide-linked dimer that activates an intrinsic tyrosine kinase activity on the M-CSF receptor by inducing dimerization of the receptor molecules. The structure of a recombinant human M-CSF dimer, determined at 2.5 angstroms by x-ray crystallography, contains two bundles of four alpha helices laid end-to-end, with an interchain disulfide bond. Individual monomers of M-CSF show a close structural similarity to the cytokines granulocyte-macrophage colony-stimulating factor and human growth hormone. Both of these cytokines are monomeric in their active form, and their specific receptors lack intrinsic tyrosine kinase activity. The similarity of these structures suggests that the receptor binding determinants for all three cytokines may be similar.
- Structural Biology Division, Lawrence Berkeley Laboratory, Berkeley, CA 94720.
Organizational Affiliation: