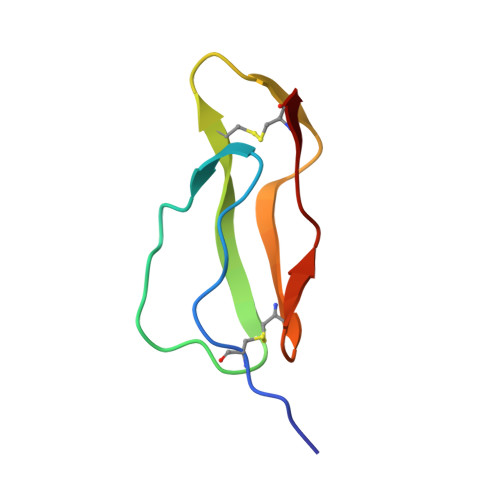
Three-dimensional structure of a complement control protein module in solution.
Norman, D.G., Barlow, P.N., Baron, M., Day, A.J., Sim, R.B., Campbell, I.D.(1991) J Mol Biology 219: 717-725
- PubMed: 1829116 Search on PubMed
- DOI: https://doi.org/10.1016/0022-2836(91)90666-t
- Primary Citation Related Structures:
1HCC - PubMed Abstract:
The complement control protein (CCP) modules (also known as short consensus repeats) are defined by a consensus sequence within a stretch of about 60 amino acid residues. These modules have been identified more than 140 times in over 20 proteins, including 12 proteins of the complement system. The solution structure of the 16th CCP module from human complement factor H has been determined by a combination of 2-dimensional nuclear magnetic resonance spectroscopy and restrained simulated annealing. In all, 548 structurally important nuclear Overhauser enhancement cross-peaks were quantified as distance restraints and, together with 41 experimentally measured angle restraints, were incorporated into a simulated annealing protocol to determine a family of closely related structures that satisfied the experimental observations. The CCP structure is shown to be based on a beta-sandwich arrangement; one face made up of three beta-strands hydrogen-bonded to form a triple-stranded region at its centre and the other face formed from two separate beta-strands. Both faces of the molecule contribute highly conserved hydrophobic side-chains to a compact core. The regions between the beta-strands are composed of both well-defined turns and less well-defined loops. Analysis of CCP sequence alignments, in light of the determined structure, reveals a high degree of conservation amongst residues of obvious structural importance, while almost all insertions, deletions or replacements observed in the known sequences are found in the less well-defined loop regions. On the basis of these observations it is postulated that models of other CCP modules that are based on the structure presented here will be accurate. Certain families of CCP modules differ from the consensus in that they contain extra cysteine residues. As a test of structural consensus, the extra disulphide bridges are shown to be easily accommodated within the determined CCP model.
- Department of Biochemistry, University of Oxford, U.K.
Organizational Affiliation: