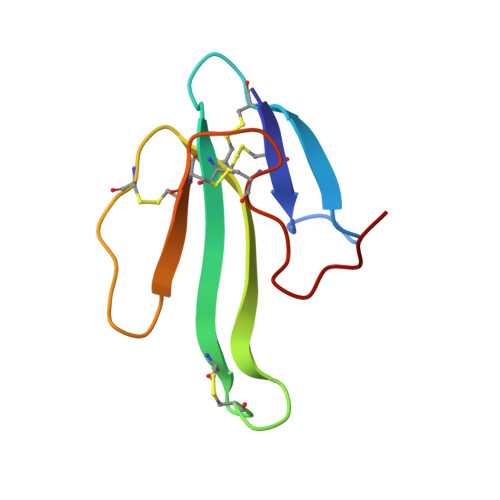
A Beta-Hairpin Structure in a 13-mer Peptide that Binds Alpha-Bungarotoxin with High Affinity and Neutralizes its Toxicity
Scherf, T., Kasher, R., Balass, M., Fridkin, M., Fuchs, S., Katchalski-Katzir, E.(2001) Proc Natl Acad Sci U S A 98: 6629
- PubMed: 11381118 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.111164298
- Primary Citation Related Structures:
1HAA, 1HAJ - PubMed Abstract:
Snake-venom alpha-bungarotoxin is a member of the alpha-neurotoxin family that binds with very high affinity to the nicotinic acetylcholine receptor (AChR) at the neuromuscular junction. The structure of the complex between alpha-bungarotoxin and a 13-mer peptide (WRYYESSLEPYPD) that binds the toxin with high affinity, thus inhibiting its interactions with AChR with an IC(50) of 2 nM, has been solved by (1)H-NMR spectroscopy. The bound peptide folds into a beta-hairpin structure created by two antiparallel beta-strands, which combine with the already existing triple-stranded beta-sheet of the toxin to form a five-stranded intermolecular, antiparallel beta-sheet. Peptide residues Y3(P), E5(P), and L8(P) have the highest intermolecular contact area, indicating their importance in the binding of alpha-bungarotoxin; W1(P), R2(P), and Y4(P) also contribute significantly to the binding. A large number of characteristic hydrogen bonds and electrostatic and hydrophobic interactions are observed in the complex. The high-affinity peptide exhibits inhibitory potency that is better than any known peptide derived from AChR, and is equal to that of the whole alpha-subunit of AChR. The high degree of sequence similarity between the peptide and various types of AChRs implies that the binding mode found within the complex might possibly mimic the receptor binding to the toxin. The design of the high-affinity peptide was based on our previous findings: (i) the detection of a lead peptide (MRYYESSLKSYPD) that binds alpha-bungarotoxin, using a phage-display peptide library, (ii) the information about the three-dimensional structure of alpha-bungarotoxin/lead-peptide complex, and (iii) the amino acid sequence analysis of different AChRs.
- Department of Chemical Services, The Weizmann Institute of Science, Rehovot 76100, Israel.
Organizational Affiliation: