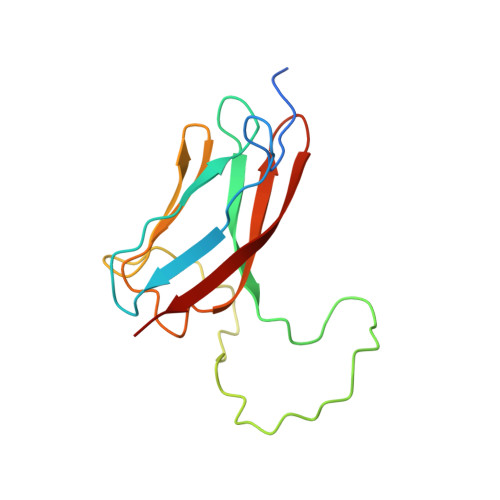
Structure, Stability and Dynamics of the Central Domain of Cardiac Myosin Binding Protein C (Mybp-C): Implications for Multidomain Assembly and Causes for Cardiomyopathy
Idowu, S., Gautel, M., Perkins, S., Pfuhl, M.(2003) J Mol Biology 329: 745
- PubMed: 12787675 Search on PubMed
- DOI: https://doi.org/10.1016/s0022-2836(03)00425-x
- Primary Citation Related Structures:
1GXE - PubMed Abstract:
The large multidomain muscle protein myosin binding protein C (MyBP-C) has been implicated for some time in cardiac disease while until recently little was known about its structure and function. Here we present a detailed study of the central domain C5 of the cardiac isoform of MyBP-C. This domain is unusual in several aspects. Firstly it contains two sizeable insertions compared to the non-cardiac isoforms. The first insertion comprises the linker between domains cC4 and cC5 that is elongated by ten amino acid residues, the second insertion comprises an elongation of the CD-loop in the middle of the domain by approximately 30 amino acid residues. Secondly two point mutations linked to familial hypertrophic cardiomyopathy (FHC) have been identified in this domain. This work shows that the general fold of cC5 is in agreement with the IgI family of beta-sandwich structures. The long cardiac-specific linker between cC4 and cC5 is not a linker at all but an integral part of the fold of cC5, as evidenced by an unfolded mutant in which this segment was removed. The second insertion is shown to be unstructured, highly dynamic and mostly extended according to NMR relaxation measurements and analytical ultracentrifugation. The loss of several key interactions conserved in the CD-loop of the IgI fold is assumed to be responsible for the low stability of cC5 compared to other IgI domains from titin and MyBP-C itself. The low thermodynamic stability of cC5 is most evident in one of the two FHC-linked mutations, N755K (Asn115 in this construct) which is mainly unfolded with a small proportion of a native-like folded species. In contrast, the second FHC-linked mutation, R654H (Arg14 in this construct) is as well folded and stable as the wild-type. This residue is located in the extended beta-bulge at the N terminus of the protein, pointing towards the surface of the CFGA' beta-sheet. This position is in agreement with recent data pointing to a function of Arg654 in an intermolecular interaction with MyBP-C domain cC8.
- Department of Pharmacology, University College London, Gower Street, WC1E 6BT, London, UK.
Organizational Affiliation: