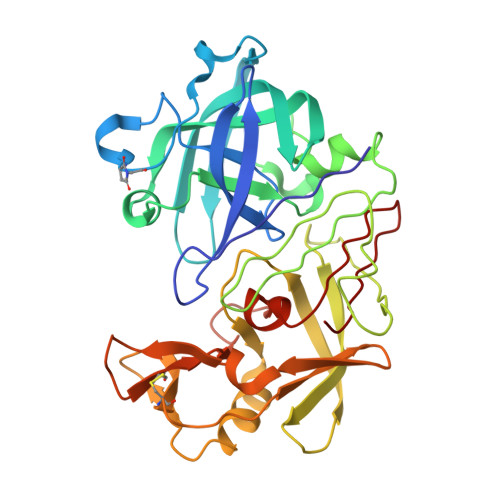
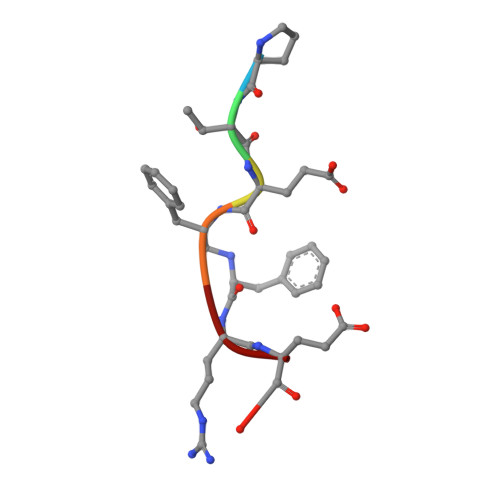
Five Atomic Resolution Structures of Endothiapepsin Inhibitor Complexes: Implications for the Aspartic Proteinase Mechanism
Coates, L., Erskine, P.T., Crump, M.P., Wood, S.P., Cooper, J.B.(2002) J Mol Biology 318: 1405
- PubMed: 12083527 Search on PubMed
- DOI: https://doi.org/10.1016/s0022-2836(02)00197-3
- Primary Citation Related Structures:
1GVT, 1GVU, 1GVV, 1GVW, 1GVX - PubMed Abstract:
Endothiapepsin is derived from the fungus Endothia parasitica and is a member of the aspartic proteinase class of enzymes. This class of enzyme is comprised of two structurally similar lobes, each lobe contributing an aspartic acid residue to form a catalytic dyad that acts to cleave the substrate peptide bond. The three-dimensional structures of endothiapepsin bound to five transition state analogue inhibitors (H189, H256, CP-80,794, PD-129,541 and PD-130,328) have been solved at atomic resolution allowing full anisotropic modelling of each complex. The active sites of the five structures have been studied with a view to studying the catalytic mechanism of the aspartic proteinases by locating the active site protons by carboxyl bond length differences and electron density analysis. In the CP-80,794 structure there is excellent electron density for the hydrogen on the inhibitory statine hydroxyl group which forms a hydrogen bond with the inner oxygen of Asp32. The location of this proton has implications for the catalytic mechanism of the aspartic proteinases as it is consistent with the proposed mechanism in which Asp32 is the negatively charged aspartate. A number of short hydrogen bonds (approximately 2.6 A) with ESD values of around 0.01 A that may have a role in catalysis have been identified within the active site of each structure; the lengths of these bonds have been confirmed using NMR techniques. The possibility and implications of low barrier hydrogen bonds in the active site are considered.
- School of Biological Sciences, University of Southampton, UK. leightonc@bigfoot.com
Organizational Affiliation: