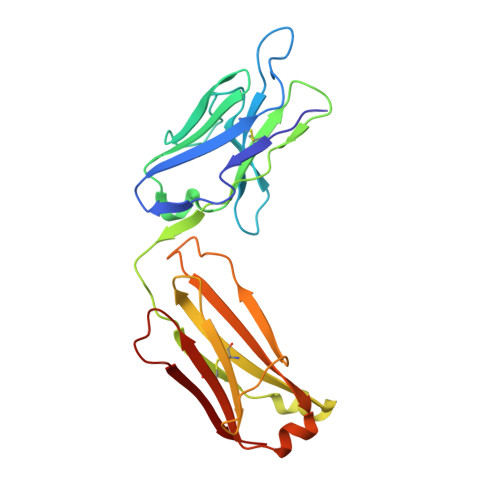
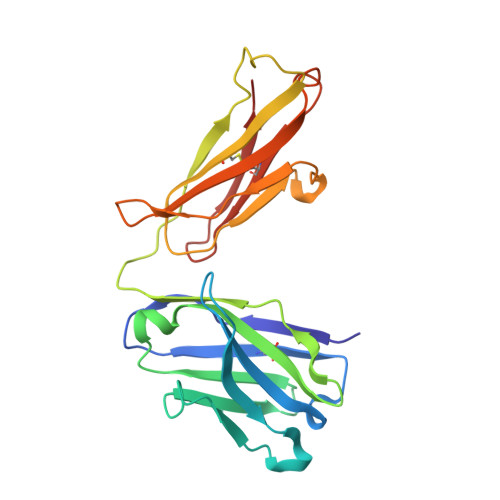
A comparative analysis of the immunological evolution of antibody 28B4.
Yin, J., Mundorff, E.C., Yang, P.L., Wendt, K.U., Hanway, D., Stevens, R.C., Schultz, P.G.(2001) Biochemistry 40: 10764-10773
- PubMed: 11535051 Search on PubMed
- DOI: https://doi.org/10.1021/bi010536c
- Primary Citation Related Structures:
1FL5, 1FL6 - PubMed Abstract:
In an effort to gain greater insight into the evolution of the redox active, catalytic antibody 28B4, the germline genes used by the mouse to generate this antibody were cloned and expressed, and the X-ray crystal structures of the unliganded and hapten-bound germline Fab of antibody 28B4 were determined. Comparison with the previously determined structures of the unliganded and hapten-bound affinity-matured Fab [Hsieh-Wilson, L. C., Schultz, P. G., and Stevens, R. C. (1996) Proc. Natl. Acad. Sci. U.S.A. 93, 5363] shows that the germline antibody binds the p-nitrophenyl ring of hapten 3 in an orientation significantly different from that seen in the affinity-matured antibody, whereas the phosphonate moiety is bound in a similar mode by both antibodies. The affinity-matured antibody 28B4 has more electrostatic and hydrophobic interactions with hapten 3 than the germline antibody and binds the hapten in a lock-and-key fashion. In contrast, significant conformational changes occur in the loops of CDR H3 and CDR L1 upon hapten binding to the germline antibody, consistent with the notion of structural plasticity in the germline antibody-combining site [Wedemayer, G. J., Patten, P. A., Wang, L. H., Schultz, P. G., and Stevens, R. C. (1997) Science 276, 1665]. The structural differences are reflected in the differential binding affinities of the germline Fab (K(d) = 25 microM) and 28B4 Fab (K(d) = 37 nM) to hapten 3. Nine replacement mutations were found to accumulate in the affinity-matured antibody 28B4 compared to its germline precursor. The effects of each mutation on the binding affinity of the antibody to hapten 3 were characterized in detail in the contexts of both the germline and the affinity-matured antibodies. One of the mutations, Asp95(H)Trp, leads to a change in the orientation of the bound hapten, and its presence is a prerequisite for other somatic mutations to enhance the binding affinity of the germline antibody for hapten 3. Thus, the germline antibody of 28B4 acquired functionally important mutations in a stepwise manner, which fits into a multicycle mutation, affinity selection, and clonal expansion model for germline antibody evolution. Two other antibodies, 20-1 and NZA6, with very different antigen specificities were found to be highly homologous to the germline antibody of 28B4, consistent with the notion that certain germline variable-region gene combinations can give rise to polyspecific hapten binding sites [Romesberg, F. E., Spiller, B., Schultz, P. G., and Stevens, R. C. (1998) Science 279, 1929]. The ultimate specificity of the polyspecific germline antibody appears to be defined by CDR H3 variability and subsequent somatic mutation. Insights into the evolution of antibody-combining sites provided by this and other structural studies are discussed.
- Department of Chemistry and Molecular Biology, The Scripps Research Institute, 10550 North Torrey Pines Road, La Jolla, California 92037, USA.
Organizational Affiliation: