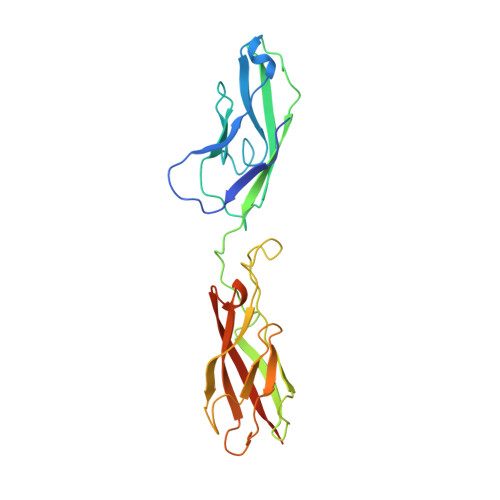
Structural basis of calcium-induced E-cadherin rigidification and dimerization.
Nagar, B., Overduin, M., Ikura, M., Rini, J.M.(1996) Nature 380: 360-364
- PubMed: 8598933 Search on PubMed
- DOI: https://doi.org/10.1038/380360a0
- Primary Citation Related Structures:
1EDH - PubMed Abstract:
The cadherins mediate cell adhesion and play a fundamental role in normal development. They participate in the maintenance of proper cell-cell contacts: for example, reduced levels of epithelial cadherin (E-cadherin) correlate with increased invasiveness in many human tumour cell types. The cadherins typically consist of five tandemly repeated extracellular domains, a single membrane-spanning segment and a cytoplasmic region. The N-terminal extracellular domains mediate cell-cell contact while the cytoplasmic region interacts with the cytoskeleton through the catenins. Cadherins depend on calcium for their function: removal of calcium abolishes adhesive activity, renders cadherins vulnerable to proteases (reviewed in ref. 4) and, in E-cadherin, induces a dramatic reversible conformational change in the entire extracellular region. We report here the X-ray crystal structure at 2.0 A resolution of the two N-terminal extracellular domains of E-cadherin in the presence of calcium. The structure reveals a two-fold symmetric dimer, each molecule of which binds a contiguous array of three bridged calcium ions. Not only do the bound calcium ions linearize and rigidify the molecule, they promote dimerization. Although the N-terminal domain of each molecule in the dimer is aligned in a parallel orientation, the interactions between them differ significantly from those found in the neural cadherin (N-cadherin) N-terminal domain (NCD1) structure. The E-cadherin dual-domain structure reported here defines the role played by calcium in the cadherin-mediated formation and maintenance of solid tissues.
- Department of Molecular and Medical Genetics, University of Toronto, Ontario, Canada.
Organizational Affiliation: