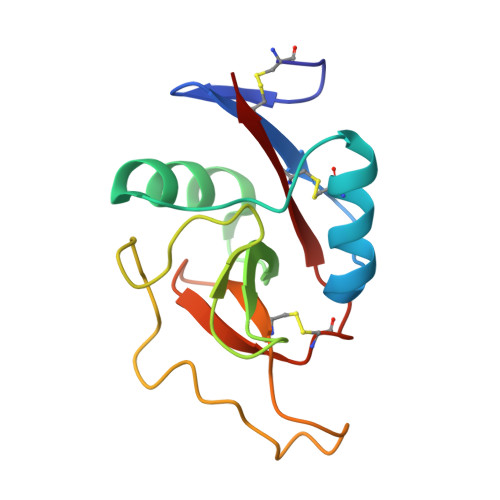
Crystal structure of the carbohydrate recognition domain of the H1 subunit of the asialoglycoprotein receptor.
Meier, M., Bider, M.D., Malashkevich, V.N., Spiess, M., Burkhard, P.(2000) J Mol Biology 300: 857-865
- PubMed: 10891274 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.2000.3853
- Primary Citation Related Structures:
1DV8 - PubMed Abstract:
The human asialoglycoprotein receptor (ASGPR), also called hepatic lectin, is an integral membrane protein and is responsible for the clearance of desialylated, galactose-terminal glycoproteins from the circulation by receptor-mediated endocytosis. It can be subdivided into four functional domains: the cytosolic domain, the transmembrane domain, the stalk and the carbohydrate recognition domain (CRD). The galactose-binding domains belong to the superfamily of C-type (calcium-dependent) lectins, in particular to the long-form subfamily with three conserved intramolecular disulphide bonds. It is able to bind terminal non-reducing galactose residues and N-acetyl-galactosamine residues of desialated tri or tetra-antennary N-linked glycans. The ASGPR is a potential liver-specific receptor for hepatitis B virus and Marburg virus and has been used to target exogenous molecules specifically to hepatocytes for diagnostic and therapeutic purposes.Here, we present the X-ray crystal structure of the carbohydrate recognition domain of the major subunit H1 at 2.3 A resolution. While the overall fold of this and other known C-type lectin structures are well conserved, the positions of the bound calcium ions are not, indicating that the fold is stabilised by alternative mechanisms in different branches of the C-type lectin family. It is the first CRD structure where three calcium ions form an intergral part of the structure. In addition, the structure provides direct confirmation for the conversion of the ligand-binding site of the mannose-binding protein to an asialoglycoprotein receptor-like specificity suggested by Drickamer and colleagues. In agreement with the prediction that the coiled-coil domain of the ASGPR is separated from the CRD and its N-terminal disulphide bridge by several residues, these residues are indeed not alpha-helical, while in tetranectin they form an alpha-helical coiled-coil.
- M.E. Müller Institute for Structural Biology, University of Basel, Klingelbergstrasse 70 CH-4056 Basel, Switzerland.
Organizational Affiliation: