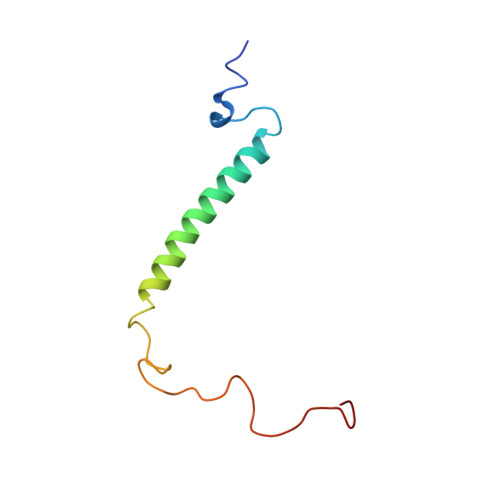
Solution structure of porcine delta sleep-inducing peptide immunoreactive peptide A homolog of the shortsighted gene product.
Seidel, G., Adermann, K., Schindler, T., Ejchart, A., Jaenicke, R., Forssmann, W.G., Rosch, P.(1997) J Biological Chem 272: 30918-30927
- PubMed: 9388238 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.272.49.30918
- Primary Citation Related Structures:
1DIP - PubMed Abstract:
The 77-residue delta sleep-inducing peptide immunoreactive peptide (DIP) is a close homolog of the Drosophila melanogaster shortsighted gene product. Porcine DIP (pDIP) and a peptide containing a leucine zipper-related partial sequence of pDIP, pDIP(9-46), was synthesized and studied by circular dichroism and nuclear magnetic resonance spectroscopy in combination with molecular dynamics calculations. Ultracentrifugation, size exclusion chromatography, and model calculations indicated that pDIP forms a dimer. This was confirmed by the observation of concentration-dependent thermal folding-unfolding transitions. From CD spectroscopy and thermal folding-unfolding transitions of pDIP(9-46), it was concluded that the dimerization of pDIP is a result of interaction between helical structures localized in the leucine zipper motif. The three-dimensional structure of the protein was determined with a modified simulated annealing protocol using experimental data derived from nuclear magnetic resonance spectra and a modeling approach based on an established strategy for coiled coil structures. The left-handed super helical structure of the leucine zipper type sequence resulting from the modeling approach is in agreement with known leucine zipper structures. In addition to the hydrophobic interactions between the amino acids at the heptade positions a and d, the structure of pDIP is stabilized by the formation of interhelical i to i' + 5 salt bridges. This result was confirmed by the pH dependence of the thermal-folding transitions. In addition to the amphipatic helix of the leucine zipper, a second helix is formed in the NH2-terminal part of pDIP. This helix exhibits more 310-helix character and is less stable than the leucine zipper helix. For the COOH-terminal region of pDIP no elements of regular secondary structure were observed.
- Lehrstuhl für Biopolymere, Universität Bayreuth, Universitätsstrasse 30, D-95447 Bayreuth, Germany.
Organizational Affiliation: