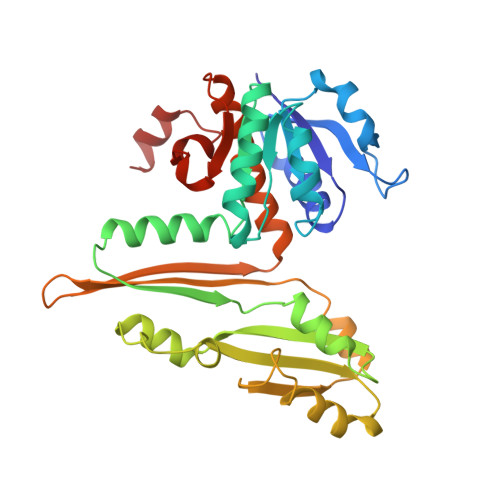
Three-dimensional structure of meso-diaminopimelic acid dehydrogenase from Corynebacterium glutamicum.
Scapin, G., Reddy, S.G., Blanchard, J.S.(1996) Biochemistry 35: 13540-13551
- PubMed: 8885833 Search on PubMed
- DOI: https://doi.org/10.1021/bi961628i
- Primary Citation Related Structures:
1DAP - PubMed Abstract:
Diaminopimelate dehydrogenase catalyzes the NADPH-dependent reduction of ammonia and L-2-amino-6-ketopimelate to form meso-diaminopimelate, the direct precursor of L-lysine in the bacterial lysine biosynthetic pathway. Since mammals lack this metabolic pathway inhibitors of enzymes in this pathway may be useful as antibiotics or herbicides. Diaminopimelate dehydrogenase catalyzes the only oxidative deamination of an amino acid of D configuration and must additionally distinguish between two chiral amino acid centers on the same symmetric substrate. The Corynebacterium glutamicum enzyme has been cloned, expressed in Escherichia coli, and purified to homogeneity using standard biochemical procedures [Reddy, S. G., Scapin, G., & Blanchard, J. S. (1996) Proteins: Structure, Funct. Genet. 25, 514-516]. The three-dimensional structure of the binary complex of diaminopimelate dehydrogenase with NADP+ has been solved using multiple isomorphous replacement procedures and noncrystallographic symmetry averaging. The resulting model has been refined against 2.2 A diffraction data to a conventional crystallographic R-factor of 17.0%. Diaminopimelate dehydrogenase is a homodimer of structurally not identical subunits. Each subunit is composed of three domains. The N-terminal domain contains a modified dinucleotide binding domain, or Rossman fold (six central beta-strands in a 213456 topology surrounded by five alpha-helices). The second domain contains two alpha-helices and three beta-strands. This domain is referred to as the dimerization domain, since it is involved in forming the monomer--monomer interface of the dimer. The third or C-terminal domain is composed of six beta-strands and five alpha-helices. The relative position of the N- and C-terminal domain in the two monomers is different, defining an open and a closed conformation that may represent the enzyme's binding and active state, respectively. In both monomers the nucleotide is bound in an extended conformation across the C-terminal portion of the beta-sheet of the Rossman fold, with its C4 facing the C-terminal domain. In the closed conformer two molecules of acetate have been refined in this region, and we postulate that they define the DAP binding site. The structure of diaminopimelate dehydrogenase shows interesting similarities to the structure of glutamate dehydrogenase [Baker, P. J., Britton, K. L., Rice, D. W., Rob, A., & Stillmann, T.J. (1992a) J. Mol. Biol. 228, 662-671] and leucine dehydrogenase [Baker, P.J., Turnbull, A.P., Sedelnikova, S.E., Stillman, T. J., & Rice, D. W. (1995) Structure 3, 693-705] and also resembles the structure of dihydrodipicolinate reductase [Scapin, G., Blanchard, J. S., & Sacchettini, J. C. (1995) Biochemistry 34, 3502-3512], the enzyme immediately preceding it in the diaminopimelic acid/lysine biosynthetic pathway.
- Department of Biochemistry, Albert Einstein College of Medicine, Bronx, New York 10461, USA.
Organizational Affiliation: