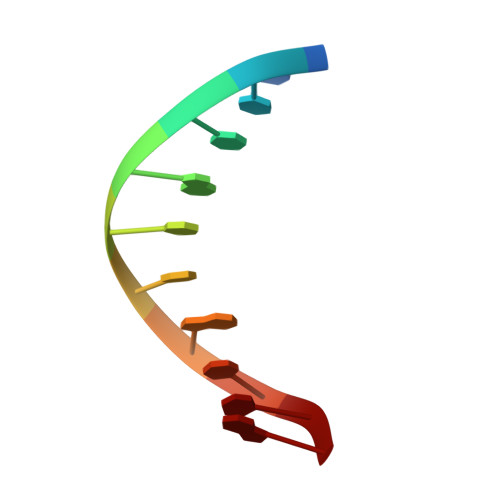
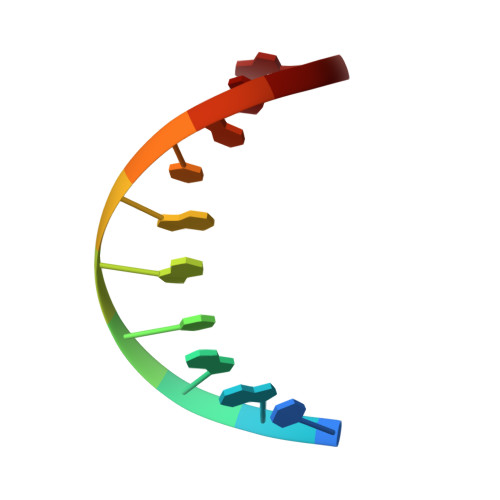
Solution structure of phage lambda half-operator DNA by use of NMR, restrained molecular dynamics, and NOE-based refinement.
Baleja, J.D., Pon, R.T., Sykes, B.D.(1990) Biochemistry 29: 4828-4839
- PubMed: 2141998 Search on PubMed
- DOI: https://doi.org/10.1021/bi00472a012
- Primary Citation Related Structures:
1D20 - PubMed Abstract:
The structure of a DNA decamer comprising the left half of the OR3 operator from bacteriophage lambda is determined in solution by using nuclear magnetic resonance spectroscopy and restrained molecular mechanics calculations. Nuclear magnetic resonance assignments for nonexchangeable protons are obtained by two-dimensional correlated and nuclear Overhauser effect (NOE) spectroscopies. Exchangeable proton resonances are assigned by one-dimensional NOE experiments. Coupling constant measurements from one- and two-dimensional experiments are used to determine approximate dihedral angles within the deoxyribose ring. Distances between protons are estimated by extrapolating distances derived from the time-dependent NOE intensities to initial mixing times. The sets of dihedral angles and distances form a basis for structure determination by restrained molecular dynamics. Separate runs start from classical A and from B DNA and converge to essentially identical structures (atomic root mean square difference of 0.8 A). The structures are improved by NOE-based refinement in which observed NOE intensities are compared to those calculated by using a full matrix analysis procedure. Final NOE residual (R) factors were less than 0.19. The resultant structures are generally B type in character, but display local sequence-dependent variations in dihedral angles and in the spatial arrangement of adjacent base pairs. Although the entire structure exhibits a small bend, the central core of the half-operator, which comprises the sequence-specific recognition site for cro repressor, is straight.
- Department of Biochemistry, University of Alberta, Edmonton, Canada.
Organizational Affiliation: