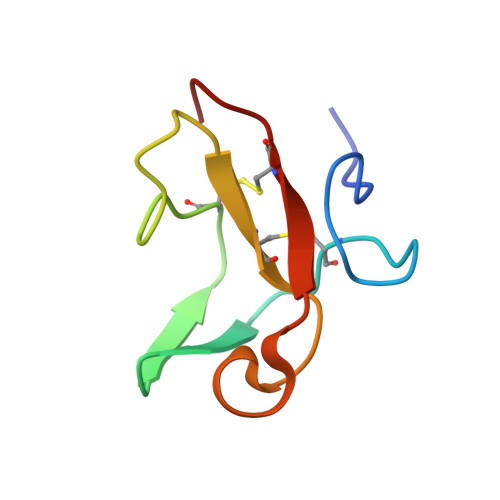
The second type II module from human matrix metalloproteinase 2: structure, function and dynamics.
Briknarova, K., Grishaev, A., Banyai, L., Tordai, H., Patthy, L., Llinas, M.(1999) Structure 7: 1235-1245
- PubMed: 10545322 Search on PubMed
- DOI: https://doi.org/10.1016/s0969-2126(00)80057-x
- Primary Citation Related Structures:
1CXW - PubMed Abstract:
Matrix metalloproteinase 2 (MMP-2, gelatinase A, 72 kDa type IV collagenase) has an important role in extracellular matrix degradation during cell migration and tissue remodeling. It is involved in development, inflammation, wound healing, tumor invasion, metastasis and other physiological and pathological processes. The enzyme cleaves several types of collagen, elastin, fibronectin and laminin. Binding to collagen is mediated by three repeats homologous to fibronectin type II modules, which are inserted in the catalytic domain in proximity to the active site. We have determined the NMR solution structure of the second type II module from human MMP-2 (col-2). The module exhibits a typical type II fold with two short double-stranded antiparallel beta sheets and three large loops packed around a cluster of conserved aromatic residues. Backbone amide dynamics, derived from (15)N relaxation experiments, correlate well with solvent accessibility and intramolecular hydrogen bonding. A synthetic peptide with the collagen consensus sequence, (Pro-Pro-Gly)(6), is shown to interact with the module. Spectral perturbations induced by (Pro-Pro-Gly)(6) binding reveal the region involved in the interaction of col-2 with collagen. The binding surface comprises exposed aromatic residues Phe21, Tyr38, Trp40, Tyr47, Tyr53 and Phe55, and the neighboring Gly33-Gly37 segment.
- Department of Chemistry Carnegie Mellon University, 4400 Fifth Avenue, Pittsburgh, PA 15213, USA.
Organizational Affiliation: